Home » Atypicals for Bipolar: And Then There Were Five
Atypicals for Bipolar: And Then There Were Five
February 1, 2005
From The Carlat Psychiatry Report
Daniel Carlat, MD
Dr. Carlat has disclosed that he has no significant relationships with or financial interests in any commercial companies pertaining to this educational activity.
We knew it was about to happen. We just didn’t know it would happen all at once. Between July and September of 2004, Seroquel, Geodon, and Abilify sequentially won approval for the treatment of manic episodes in bipolar disorder. Which means that now all of the newer atypical antipsychotics have been admitted to the bipolar club, which until December 2003 (Risperdal’s approval date), was monopolized by Zyprexa.
To help you cut through the confusion, here’s a comparison chart updating you on what each drug is or isn’t approved for:
Some comments on the chart:
General PsychiatryTo help you cut through the confusion, here’s a comparison chart updating you on what each drug is or isn’t approved for:
Table: Atypical antipsychotics approved for bipolar disorder (as of Feb 2005)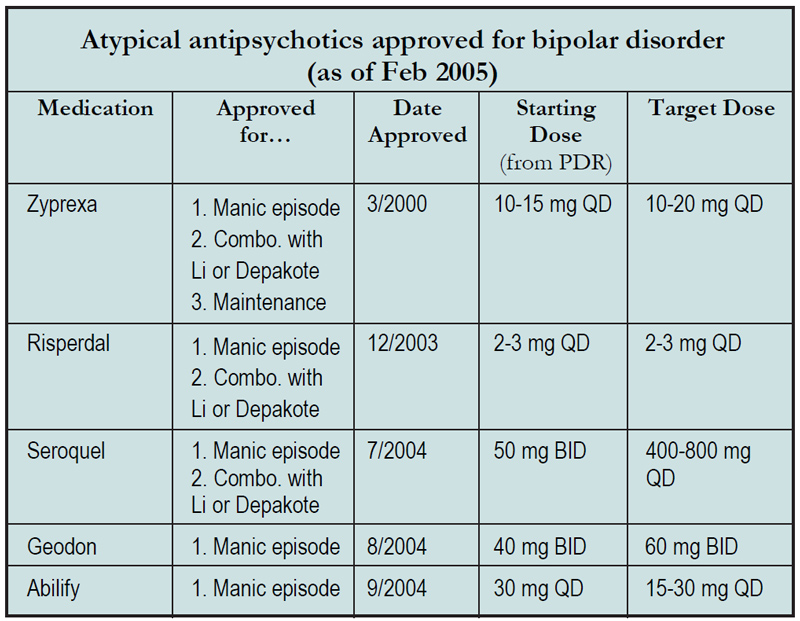
Some comments on the chart:
- You’ll notice that all five are approved for the acute treatment of a manic (and also mixed) episode. These approvals are all for monotherapy at the doses listed in the chart, and effectiveness was established with double-blind, placebo-controlled trials of three weeks duration. As an aside, conventional antipsychotics have had well-established efficacy as monotherapy for manic episodes for years, although only Thorazine applied for and received FDA approval for this.
- Three of the atypicals, Zyprexa, Risperdal, and Seroquel, are approved for "combination treatment with Lithium or Depakote.” What does this mean, exactly? Patients with bipolar disorder who were in the midst of a manic or mixed episode were recruited for these trials. They had all been on lithium or Depakote for at least two weeks, and yet they were still manic. They were then randomly divided into two groups: one group was given adjunctive antipsychotic, and the other group was given placebo. The primary outcome was improvement in the Young Mania Rating Scale (YMRS). The trials were typically three weeks in length, and at endpoint the groups on adjunctive antipsychotics had a lower average score on the YMRS than the groups taking adjunctive placebo. Thus, these studies prove the not-exactlyastonishing fact that manic patients improve more quickly when you add an antipsychotic to their mood stabilizer.
- Zyprexa has approval for one thing that none of the other atypicals can claim: “maintenance monotherapy.” This labeling was based on one unpublished study in which 361 bipolar patients who had responded to Zyprexa during an initial two week open-label treatment phase were then randomized to either continued Zyprexa or placebo. After one year, only about 20% of Zyprexa-treated patients had relapsed vs. about 60% of those on placebo. You should note, however, that we obtained this information from the Zyprexa website* and the presentation there was a very brief summary without the details needed to evaluate methodology. Nonetheless, their study methods passed FDA muster, which is usually a good sign. Whether similar maintenance data exists for the other atypicals is unknown at this point. No double-blind trials have been published, but long-term open label data exists for some atypicals (see Current Psychiatry April 2004 for a brief review).
- The chart doesn’t include Symbyax, which is a combination of Zyprexa and Prozac and is the only medication approved for the treatment of depressive episodes in bipolar disorder. We’ll review this in a future issue on bipolar disorder.
- A word on dosing. You’ll notice that the starting doses of the newer atypicals are rather high, certainly higher than doses recommended for patients with schizophrenia. This likely reflects the ardent desires of manufacturers to prove their drugs work, although less cynically, one can argue that there is indeed more urgency in treating an acute manic episode than in treating a chronic condition such as schizophrenia. But clinically, these high starting doses have led psychiatrists to cause a variety of side effects in their patients. For example, many a patient will keel over into longterm slumber-land when confronted with Geodon 40 mg BID (which is Pfizer’s recommended starting dose), while other patients will dance the akathisia jig when started on Abilify 30 mg QD (BMS’s recommendation). In fact, the Abilify package insert seems to backtrack on the 30 mg starting dose, declaring that 15% of patients had to be decreased to 15 mg QD because of tolerability issues. The bottom line is that PDR recommended dosages for bipolar disorder are often unrealistically high. On the other hand, clinicians have learned to take Seroquel’s package insert seriously, finding that you really have to push Seroquel to the 400 to 800 mg range to get a good response. Just advise your patients to invest in a good Sealy Posturepedic for the first week or so, because that’s where they’ll be spending most of their time.
KEYWORDS bipolar_disorder

Issue Date: February 1, 2005
Table Of Contents
Recommended
Newsletters
Please see our Terms and Conditions, Privacy Policy, Subscription Agreement, Use of Cookies, and Hardware/Software Requirements to view our website.
© 2026 Carlat Publishing, LLC and Affiliates, All Rights Reserved.


_-The-Breakthrough-Antipsychotic-That-Could-Change-Everything.webp?t=1729528747)



