Medications for Childhood PTSD: It’s All Off-Label
Elizabeth Tien, MD. Child, Adolescent and Adult Psychiatrist with Maimonides Medical Center in Brooklyn, NY.
Dr. Tien has declared that she has no relevant financial or other interest in any commercial companies pertaining to this activity.
In 2011, we wrote a review of medications for PTSD in children and found, unfortunately, that data to inform true evidence-based practice was scant. There are still no psychotropic medications that have been proven effective in randomized controlled trials for children with PTSD and there are no FDA approved medications for this population.
Nonetheless, these patients are suffering, and while psychotherapy is the first line treatment, it doesn’t always work, and it may not even be available to many. In the absence of compelling evidence for particular medications, we still must do what we can, and to be as rational in our prescribing decisions as possible.
In this article, I’ll update you on some of the new research, and I will offer some very concrete advice on which medications to choose, how to dose them, and what side effects to watch for. I’ll be basing my recommendations on a trio of common sense principles. First, if a medication has been shown effective for PTSD in adults, it’s reasonable to try it in children. Second, if a medication has been approved for kids for some other, non-PTSD indication, this at least means it has a track record and so may be worth trying. Finally, all other things being equal, we should try to do no harm, and so I avoid high side effect drugs like atypical antipsychotics unless I’ve run out of options.
SSRIs
Both sertraline and paroxetine are FDA approved for PTSD in adults. Two SSRIs—sertraline and fluoxetine—have been tested for PTSD in adolescents but neither of them separated from placebo in randomized controlled trials. Citalopram was effective for adolescents in open-label trials, while no studies exist for the remaining SSRIs.
In our hospital-based clinic at Maimonides Medical Center, we start with psychotherapy for pediatric PTSD. If that doesn’t work, we will try an SSRI, usually sertraline, which can help with depression, irritability, and anxiety. We choose sertraline both because of its approval for adult PTSD and because it tends to be a less activating SSRI. We often start with 12.5–25 mg in the morning. We titrate up in 12.5–25 mg increments no more than once per week, until we see a response, but we will not go higher than 200 mg daily. GI side effects like nausea are common, though they usually resolve on their own within a few days.
For kids who can’t tolerate sertraline, or if we want to achieve a higher dose more quickly, we’ll go with citalopram, starting at 10 mg, and increasing in 10 mg increments weekly, with a target dose of 20–40 mg daily. While you can also try fluoxetine, I’ve seen more behavioral activation with this medication than the other SSRIs.
Because there’s almost no evidence for other antidepressants in childhood PTSD, we rarely prescribe SNRIs, MAOIs, TCAs, trazodone, nefazodone, or mirtazapine. We know that clinicians in other practices do favor some of these medications, and given the absence of evidence one can make a reasonable case for giving them a try empirically, particularly a drug like mirtazapine, which can theoretically be helpful for traumatized kids who can’t sleep.
Antiadrenergic Agents
A small amount of research has endorsed using antiadrenergic meds for symptoms such as hyperarousal, avoidance of triggering situations, and re-experiencing symptoms. For example, a small 2013 open-label study found that extended-release guanfacine was helpful for such symptoms (Connor DF et al, J Child Adolesc Psychopharmacol 2013; 23(4):244–51).
I have found alpha agonists, both clonidine and guanfacine, to be particularly helpful with nightmares, sleep, and overall irritability and aggressive behaviors. Both of these medications have approval for the treatment of ADHD in children, which increases my comfort in using them for other indications. Get baseline vital signs before starting alpha agonists and recheck vitals when you adjust the dose or when your patient reports hypotensive symptoms such as light-headedness. When you stop these meds, you need to taper gradually to avoid rebound hypertension.
I usually start with guanfacine at 0.5 mg at bedtime and increase up to 2 mg at bedtime to treat nightmares and insomnia, although I’ve found that 1 mg is often enough. For hyperarousal symptoms, especially irritability and aggression, I’ll go with higher doses, titrating it up to 2 mg tid. The most common side effects are sedation and headaches, and sometimes hypotension, although some patients may have paradoxical increased agitation and insomnia. You can also prescribe guanfacine ER (extended release, brand name Intuniv). Start this at 1 mg a day, morning or bedtime depending on what symptoms you are trying to address, and increase in 1 mg increments on a weekly basis up to 4 mg daily. Bear in mind that there are no generics for guanfacine ER, so you’ll usually have to get a prior authorization if you want to prescribe it. Another disadvantage is that this medication can’t be crushed, chewed, or cut.
Clonidine is my second-line choice after guanfacine, primarily because families report difficulty cutting the small clonidine tabs in half. Start it at 0.05 mg at bedtime, then increase it by 0.05 mg increments every 3–7 days, up to a total dose of 0.2 mg tid. Or, you can prescribe extended-release clonidine ER (Kapvay), starting at 0.1 mg qhs and increasing by 0.1 mg a day on a weekly basis, up to a maximum dose of 0.4 mg a day. Side effects are similar to guanfacine—sedation, headaches, and possible rebound hypertension. There is currently no generic form of clonidine ER.
Beta blockers are another option, though the evidence for their use in children is pretty scant. An older open-label study of 11 cases (Famularo R et al, Am J Dis Child 1988; 142(11):1244–7) found improved symptoms of PTSD with the use of propranolol. More recent studies have tested whether the drug can prevent PTSD after exposure to trauma, but the results in children were unimpressive (Nugent NR et al, J Trauma Stress 2010; 23:282–7). Adult studies often start with 40 mg a day which is then increased up to 60–80 mg bid. Child studies start with a weight-based dose of 1–2.5 mg/kg/day up to a maximum dose of 40mg bid. Prazosin has been tried in a handful of case studies, with some efficacy. The typical starting dose in studies has been 1 mg at bedtime with an increase up to 2 mg after a week with improvements in sleep and nightmares.
Atypical Antipsychotics
While most atypical antipsychotics have pediatric indications for diagnoses ranging from bipolar disorder to psychosis to irritability in autism, I consider them a last resort in PTSD, unless there are co-morbid approved diagnoses. I favor risperidone and quetiapine, since they have shown some promising results in open-label studies of PTSD. Lower doses are usually sufficient to address hyperarousal, increased reactivity, and the aggressive and disruptive behaviors which are often what lead clinicians down the atypical antipsychotics trail.
I start risperidone at 0.25 mg a day, and increase by 0.25–0.5 mg a day, up to a dose of 3–4 mg either once daily or divided into morning and evening doses. In addition to metabolic syndrome and extrapyramidal side effects, watch for hyperprolactinemia. Start quetiapine at 25 mg a day and increase in 25–50 mg increments, up to 200 mg a day, divided bid. Sedation, dry mouth, weight gain, and GI side effects are commonly seen with quetiapine.
Mood Stabilizers
Consider mood stabilizers for traumatized youth with mood lability and aggressive behaviors that have led to hospitalization. Valproic acid and carbamazepine have been effective in open label trials. I have found valprioc acid helpful in severe emotional dysregulation, at doses titrated to serum levels in the range of 50–100 mcg/mL. The most common side effects are drowsiness, headache, and weight gain.
CCPR Verdict: Use meds only when therapy doesn’t work…then go off-label, choosing meds to target particular symptoms. Use SSRIs for depression and anxiety, alpha agonists for insomnia, nightmares, and hyperarousal. Reserve atypicals and mood stabilizers for the most severe and treatment-resistant cases.
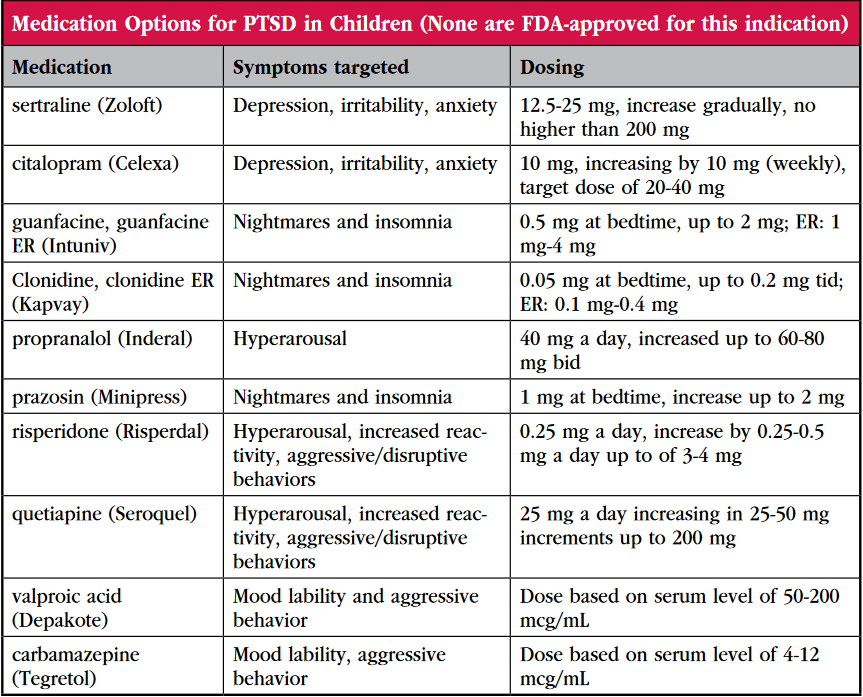 Table: Medication Options for PTSD in Children
Table: Medication Options for PTSD in Children

Newsletters
Please see our Terms and Conditions, Privacy Policy, Subscription Agreement, Use of Cookies, and Hardware/Software Requirements to view our website.
© 2026 Carlat Publishing, LLC and Affiliates, All Rights Reserved.


_-The-Breakthrough-Antipsychotic-That-Could-Change-Everything.webp?t=1729528747)



