Silexan: A Novel Anxiolytic
Patients with generalized anxiety disorder (GAD) often look to medications for help, but GAD is one of the disorders where medications are likely to disappoint. Effect sizes tend to fall in the small range (0.2–0.3) with meds for GAD, including SSRIs, SNRIs, buspirone, and pregabalin (Lyrica). Only the benzodiazepines have an effect in GAD that’s big enough for the casual observer to notice, with an effect size of 0.5 (Gomez AF et al, Expert Opin Pharmacother 2018;19(8):883–894). So when an oral extract of lavender called Silexan outperformed paroxetine in GAD with a large effect size of 0.87, it caught my attention.
What is Silexan?
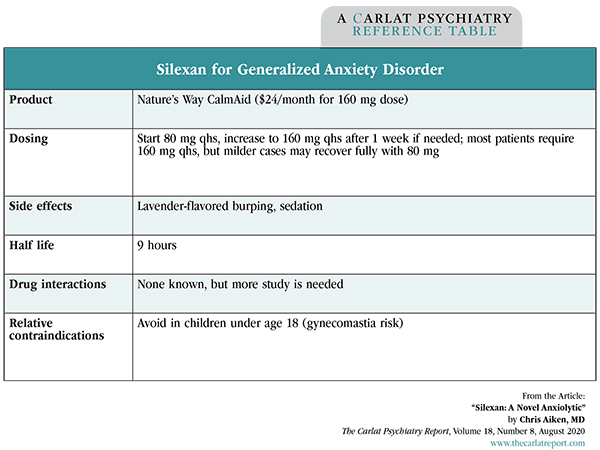
Silexan is a branded extract of lavender that was developed by Schwabe Pharmaceuticals in 2002. Its active ingredients—linalool and linalyl acetate—comprise 71% of the oil, with the remaining 29% made up of trace amounts of over a hundred other compounds from the lavender plant. Silexan is regulated as a prescription and licensed for anxiety in 14 countries. In the US, it is available over the counter as CalmAid, through Schwabe’s “Nature’s Way” line.
Silexan’s pharmacologic properties are more in line with conventional medications than with most CAM therapies. Like pregabalin, it inhibits calcium channels; like the benzos, it has GABA-ergic effects; like ketamine, it is an NMDA antagonist; and like buspirone and vortioxetine, it is a serotonin-1A agonist. The serotonin-1A agonism is probably the most relevant mechanism, judging from animal studies where Silexan’s anxiolytic effects were measured after blocking the various mechanisms (Baldinger P et al, Int J Neuropsychopharmacol 2014;18(4):pyu063).
The research
The best evidence of Silexan’s benefit in GAD comes from a large (n = 539) randomized, double-blind controlled trial that compared two doses of Silexan (80 mg or 160 mg/day) to paroxetine 20 mg/day and placebo. This 10-week study had four important outcomes:
- Both doses of Silexan worked significantly better than paroxetine and placebo (p < 0.01) on the primary outcome (Hamilton Anxiety Rating Scale) as well as on secondary measures of depression, sleep quality, and quality of life.
- The effects were dose dependent, with an effect size of 0.4 for the 80 mg dose and 0.5 for the 160 mg dose.
- Half of the patients on the 160 mg dose achieved remission.
- Silexan was as well tolerated as placebo.
Three smaller controlled trials have tested Silexan in GAD, mixed anxiety-depression, and subsyndromal anxiety. A Cochrane-style meta-analysis of all the trials found a medium effect size (0.67) for anxiety and a large effect (0.87) in studies that exclusively enrolled patients with GAD (Generoso MB et al, J Clin Psychopharmacol 2017;37(1):115–117). One of those studies found that low-dose Silexan (80 mg/day) was comparable to low-dose lorazepam (0.5 mg/day) after 6 weeks in patients with GAD.
The strengths of these studies include double blinding, intent-to-treat analysis, and restriction of adjunctive medications or psychotherapies. On the other hand, most were manufacturer funded and came from a single academic center (University of Vienna).
Silexan’s benefits are usually apparent after 2 weeks, and they continue to build over the next 3 months. Although not directly sedating, Silexan does improve sleep quality, an effect that appears to be secondary to its anxiolytic properties (Seifritz E et al, J Psychiatr Res 2019;115:69–74).
Safety
Silexan has no known withdrawal effects, addictive potential, or drug interactions. Burping was the only side effect that emerged in the clinical trials of 1165 subjects. In animal models, lavender oil is neuroprotective (Sánchez-Vidaña DI et al, Neurosci Lett 2019;701:180–192).
Lavender oil is considered safe for consumption by the FDA and NIH, with one exception. Some of the ingredients have weak estrogen-like properties, and there are rare cases of breast development in adolescent boys and early breast development in young girls who were exposed to lavender oils or aromatherapy (Ramsey JT et al, J Clin Endocrinol Metab 2019;104(11):5393–5405). I would avoid it in children under age 18.
There are no reports of hormonal problems in adults, but patients with a history of estrogen-sensitive cancers should probably avoid it unless approved by their oncologist. The risk here is unclear, as lavender extracts are actually being explored as treatments for breast cancer (Al-Sheddi ES, Asian Pac J Cancer Prev 2019;20(10):2943–2949).
Aromatherapy
The aromatherapy form of lavender is what inspired the Silexan research. The plant’s oils have been used in this way to calm the nerves and induce sleep for centuries. These benefits have nothing to do with lavender’s aroma, as mice that are unable to smell respond equally well to its anxiolytic effects. My hunch is that the lavender mist delivers the same active compounds as Silexan through the intranasal route. Lavender oil can also be rubbed on the skin, but this method is not as effective as the aromatherapy. Based on indirect comparisons, aromatherapy works faster than oral Silexan, but Silexan is more potent after several weeks of treatment (Sayed AM, Gen Hosp Psychiatry 2020;64:33–40).
How to use Silexan
Silexan is a reasonable choice for patients who have not responded to FDA-approved treatments for GAD. Patients who prefer natural approaches could use it first-line. For others, the natural aspect will invite skepticism, and they may benefit from a discussion of Silexan’s pharmacologic properties.
I started recommending Silexan in 2016 and have treated 505 patients with it, most with GAD or a mood disorder with anxious distress. My experience has been in line with the research, with the best results in pure GAD. Most patients were taking long-term benzos, and many spontaneously reduced their benzo dose on their own after starting it. Patients often forget to reorder Silexan, and when they come off it, it usually takes a few months for the anxiety to return. The change is so gradual that many don’t notice it. This is where careful tracking with rating scales (which can be as simple as having patients rate their anxiety from 1–10) and trials on and off Silexan come in handy.
TCPR Verdict: Most CAM therapies do not have robust effects, but Silexan is an exception. Consider it in adults with generalized anxiety disorder.
Table: Silexan for Generalized Anxiety Disorder
(Click to view full-size PDF.)

Newsletters
Please see our Terms and Conditions, Privacy Policy, Subscription Agreement, Use of Cookies, and Hardware/Software Requirements to view our website.
© 2026 Carlat Publishing, LLC and Affiliates, All Rights Reserved.


_-The-Breakthrough-Antipsychotic-That-Could-Change-Everything.webp?t=1729528747)



