Management of Depression During Pregnancy and Breastfeeding
CHPR: Dr. Horst, please start by telling us how to screen for depression during pregnancy.
Dr. Horst: Best practices for screening have evolved considerably over the years, and we’ve learned that it is best to screen all women for mood disorders during pregnancy. The classic screening tool is the Edinburgh Postnatal Depression Scale (EPDS), which effectively identifies depression in both the antenatal and postpartum periods (Sit DK and Wisner KL, Clin Obstet Gynecol 2009;52(3):456–468). It is easy to administer and available in many languages. You can also use the Patient Health Questionnaire (PHQ-9) so long as you ignore the two questions that have to do with sleep and appetite. We also use the seven-item, self-reported anxiety questionnaire (GAD-7) because anxiety is such a big part of perinatal depression (Editor’s note: see “Screening Tools for Depression During Pregnancy” table on page 8).
CHPR: Why do you skip the questions for sleep and appetite on the PHQ-9?
Dr. Horst: Pregnancy is such a physically altered state that most women in the perinatal period have disrupted sleep and appetite. You’ll get false-positive screenings for depression if you don’t ignore those questions.
CHPR: That makes sense. What are the main risk factors for perinatal depression?
Dr. Horst: A personal history of a mood disorder is a primary risk factor, so anyone who has a history of depression and/or bipolar disorder (BD) is at higher risk. Patients going through stress are at risk, whether this is financial trouble, a death in the family, or an unwanted pregnancy. We also know that under-resourced people are at higher risk. So young, low-income patients with unexpected pregnancies are at greater risk. A lack of social support, particularly from their partners, also places patients at greater risk. Family history of mental health issues is another factor, particularly if it includes a family history of BD.
CHPR: Many pregnant women face those risk factors. How common is depression during pregnancy?
Dr. Horst: About 10%–15% of women experience clinically significant depressive symptoms during pregnancy (Flynn HA et al, General Hospital Psychiatry 2006;28(4):289–299).
CHPR: For patients with preexisting depression who are now stable on a maintenance antidepressant, how do you decide whether to continue the antidepressant during pregnancy?
Dr. Horst: I recommend that they remain on the antidepressant rather than risk a relapse of depression. Antidepressants are the most studied of all medications used in pregnancy, and studies have found high rates of relapse among pregnant women who discontinue antidepressants. One study, for example, found that 68% of pregnant patients relapsed to depression following antidepressant discontinuation, compared to 26% of patients who remained on the medication (Cohen L et al, JAMA 2006;295:499–507).
CHPR: It sounds like antidepressants can really help maintain mood stability during pregnancy. What do the data show regarding the risk of birth defects or neurodevelopmental outcomes following prenatal antidepressant exposure?
Dr. Horst: As a whole, studies have not found an increased risk for major malformations when antidepressants are used in the first trimester (Viswanathan M et al. Maternal, Fetal, and Child Outcomes of Mental Health Treatments in Women: A Systematic Review of Perinatal Pharmacologic Interventions. Rockville, MD: Agency for Healthcare Research and Quality; 2021). Additionally, a recent study of over 145,000 children found no increase in the rate of autism spectrum disorder, ADHD, learning disorders, or developmental and behavioral disorders following in-utero exposure to antidepressants. This study took into account factors such as genetic factors and maternal use of alcohol and tobacco (Suarez EA et al, JAMA Intern Med 2022;182(11):1149–1160).
CHPR: It sounds like the data are quite reassuring overall.
Dr. Horst: Yes, for the most part, but the data aren’t perfect: A problem with research into pregnancy outcomes is that studies consist mostly of epidemiologic examinations of databases or naturalistic studies. A lot of studies also don’t account for confounders, like maternal age and use of additional medications or substances. When we look at studies that account statistically for confounding variables, the findings are reassuring. A group of researchers led by Krista Huybrechts, for example, has published several well-controlled, large studies (Suarez EA et al, JAMA Intern Med 2022;182(11):1149–1160). So, bottom line, the data at this point show that if antidepressants help pregnant patients stay healthy, feel less stressed, sleep better, and take better care of themselves during their pregnancies, then it’s better that they stay on the medication.
CHPR: When we weigh the risks of prenatal exposure to antidepressants, it’s important to also consider the risk of untreated depressive symptoms on birth outcomes, right?
Dr. Horst: Right. The negative impacts of prenatal depressive symptoms on birth outcomes have been known for decades. They include low birth weight, preterm birth, and poor maternal health behaviors such as substance use and inadequate attendance to prenatal visits. A recent study found that prenatal depression and anxiety were linked to greater odds of adverse pregnancy outcomes than well-established risk factors like advanced maternal age and obesity (Accortt E et al, J Matern Fetal Neonatal Med 2022;35(25):9066–9070). 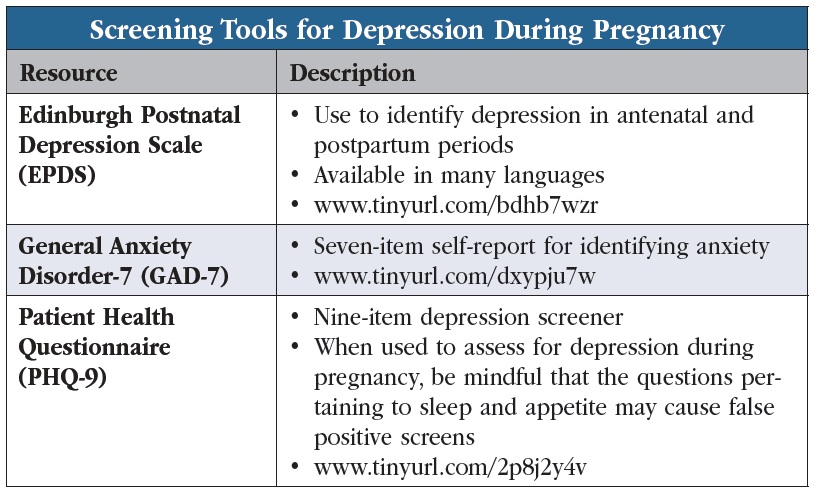
CHPR: Do newborns experience withdrawal symptoms once they’re born and are no longer exposed to the antidepressants in utero?
Dr. Horst: Clinicians have expressed concern about poor neonatal adaptation syndrome (PNAS) in babies whose mothers took antidepressants during pregnancy. Symptoms of PNAS include jitteriness, respiratory problems, altered muscle tone, and feeding problems. The evidence for this syndrome is low quality, but I still warn patients about the potential concern. Proposed causes for PNAS are either antidepressant withdrawal or serotonin toxicity. However, a study that controlled for maternal illness severity found no difference in neonatal symptoms between infants exposed to SSRIs in the last 14 days of gestation and those exposed earlier in pregnancy (Warburton W et al, Acta Psychiatry Scandinavia 2010;121(6):471–479). Unfortunately, prescription bottles for antidepressants still carry a warning about use in the third trimester of pregnancy. I address these concerns frequently when patients bring home a prescription and see this warning. I tell the moms that any PNAS symptoms are transient and mild, whereas the risk for a recurrence of depression is highest in the early postpartum period.
CHPR: After the baby is born, is it safe for patients to breastfeed while taking antidepressant medications?
Dr. Horst: We have good data, especially for the older medications, about how much gets through in breast milk. In general, medications that are more highly protein-bound and have shorter half-lives tend to be less likely to accumulate in breast milk—so medications like sertraline and citalopram, for example, can be good choices. LactMed, Reprotox, and www.mothertobaby.org provide medication safety data in breastfeeding and are great resources for clinicians. The American Academy of Pediatrics came out with recommendations saying that if the infant plasma concentration is less than 10% of the therapeutic maternal plasma concentration, it’s generally safe for the baby to be exposed to the medication, and most of our medications, including SSRIs, reach levels in the infant that are well below 10% of the mother’s levels (Sachs HC et al, Pediatrics 2013;132(3):e796–e809).
CHPR: Can you give us some tips for risk/benefit discussions with patients?
Dr. Horst: They should involve joint decision making where we learn about the mothers’ risk tolerance and values and ideally also their partners’. Then we review the data and our clinical experience. It’s a case-by-case discussion, and there are no black-and-white recommendations. Important questions to include in the risk/benefit discussion are “Have you ever discontinued medications before? If so, how ill did you get afterwards? How quickly did the relapse of symptoms occur?” It can be a challenge when patients tell us “If I stay on the medication and anything goes wrong with my pregnancy or my baby, I will never forgive myself.” But when patients have support from their family and have access to an expert who shares the latest research data supporting their recommendations, they tend to be comfortable with staying on medications. And it’s always good to encourage patients to get help through a variety of modalities of treatment, including psychotherapy, behavioral techniques including cognitive behavioral therapy, stress reduction, and psychoeducation (Li X et al, Clin Psychol Rev 2022;92:102129).
CHPR: From a medico-legal standpoint, are there any points we should be sure to include and document in our risk/benefit discussions?
Dr. Horst: In our clinic, we use this wording: “A thorough review of the available scientific literature to date was conducted, including the risks of no treatment to both the mother and the baby, and the current known risks of medication to the fetus both during pregnancy and the postpartum period. The patient was informed that no medication is approved for use during pregnancy but that in some cases the benefits of treatment to both the mother and the fetus outweigh the current known or unknown risks of medication use during pregnancy and the postpartum period. The patient states her understanding of this and has chosen to start or continue treatment.”
CHPR: What support and educational resources can we provide our patients?
Dr. Horst: I like to give patients the option to consult with www.mothertobaby.org because that provides a free second opinion. The website is operated by the Organization of Teratology Information Specialists (OTIS), experts in the field of birth defect research, and errs on the side of caution—OTIS does not make recommendations unless they’re quite comfortable with the data. Patients can chat for free with a teratology specialist in real time; they can have a phone call if they want at 866-626-6847 (www.mothertobaby.org/contact/). It’s a really good resource. For patients who are comfortable with reading research studies, I give them articles and refer them to www.womensmentalhealth.org.
CHPR: Those are all great resources. Is there anything else we should address?
Dr. Horst: I will end by saying that there is still a large gap between general psychiatric knowledge and the field of perinatal psychiatry. I frequently get referrals from patients who were told by their psychiatrists that they needed to discontinue their medications when they learned they were pregnant. By the time I see them, many patients have already relapsed after stopping their medications, so I feel passionate about educating clinicians about the treatment of psychiatric illnesses in pregnancy. I appreciate you doing this Q&A!
CHPR: Thank you for your time, Dr. Horst.
 Karen Horst, MD.
Karen Horst, MD. 

_-The-Breakthrough-Antipsychotic-That-Could-Change-Everything.webp?t=1729528747)



