How to Treat Opiate Use Disorders
Professor and medical director at the Center for Neurobehavioral Research on Addictions at the University of Texas Medical School.
Dr. Weaver has disclosed that he has no relevant financial or other interests in any commercial companies pertaining to this educational activity.
Opiate use disorder is increasingly common, and it is important to develop a systematic approach for treating it. In this article, I will discuss some of the more important elements of treatment, starting with the need to address patients’ denial and moving on to some of the nitty gritty aspects of medication-assisted treatment.
Acknowledging the problem
In asking patients about their opiate use, I have encountered many forms of denial. Here are two of the more common ones.
"My problem isn't very serious."
Even people who acknowledge that they have a problem are very good at compartmentalizing—using both ambivalence and denial. They may say, “Well, I just need help with the physical part of my addiction. If I could do a quick detox I’d be fine. I don’t need to do anything else beyond that.” But in fact, most patients are unlikely to be successful without other support and will quickly relapse.
“I don’t have a drug problem, I just use it sometimes to deal with stress.”
Self-medicating is very common. People will use what they have at hand to self-treat their depression or anxiety symptoms without recognizing they are doing so. They may use their Vicodin in the same way others would “drown their sorrows” in alcohol.
Motivational interviewing
I often use motivational interviewing techniques during my initial evaluations (for details on the approach, see our interview with Dr. William Miller in CATR, October 2014). I start by helping people to recognize that there might be a problem. This usually involves helping them to see the discrepancy between where they are now and where they would like to be. Then I help them understand some of the things that they can work on to successfully achieve their goals.
One of the main challenges is to help people acknowledge what sort of pain they are using the drugs to address—physical pain, emotional pain, or both. Patients commonly end up abusing opiates after having received a narcotic prescription for a pain issue and then continuing to use it for other reasons. I try to help them recognize that there’s a difference between emotional pain and physical pain. The medication may temporarily alleviate some symptoms of both of those things, but there are also negative aspects of using an opioid this way, and I help them to think this through.
Some patients readily admit that they use an opiate daily for emotional issues. If so, I might respond with, “Is this strategy giving you the kind of emotional stability that you want? After all, there are other ways to get through tough days. Have you really considered the potential consequences, including to your health, legal issues, and how others would perceive you for doing this sort of thing?” This will often lead to a discussion of alternative treatments, like counseling or psychiatric medications.
People often overestimate how much control they have over their use of opiates. Things may appear relatively stable from their point of view, but something unexpected may happen, causing more stress, and they escalate their use temporarily—only to realize they can’t reduce their dose back to what they were taking before. And if they keep increasing the dose, tolerance and withdrawal symptoms appear. Such symptoms may drive people to do more egregious things to obtain their fix, and they may start to recognize that this is not how they want to live their lives.
Treatment
Effective treatment of any substance use disorder involves psychosocial interventions at the least, often with the addition of medication-assisted treatment. In this section, I’ll focus on how to use medications such as methadone and buprenorphine to help your patients who are abusing opioids.
Detox
While we often think of detox as a short-term process, the official federal definition allows for up to a 6-month taper. Such a long detox can often be done on an outpatient basis. Some patients will opt for detox, while others will do better with maintenance treatment (defined as staying on a prescribed opiate for greater than 6 months). Some patients require inpatient detox, and the American Society of Addiction Medicine (ASAM) criteriacan help clinicians make that decision (see CATR, November 2015 for an entire issue explaining the ASAM criteria). In general, inpatient detox works best for patients with more severe addictions, those with psychiatric or medical issues, and those with challenging psychosocial situations, like homelessness and/or continued availability of substances at home.
Outpatient tapering
Some patients are able to gradually taper down their use of oral opioids without a specific detox protocol. For the most part, these are patients who have been able to obtain the medications legitimately. For example, patients may have started taking opioids because of some dental work, but later found that they became dependent, first psychologically then physically. They may have been obtaining it by going to multiple providers or perhaps one provider has continued to provide refills. In this context, it would be reasonable to try a therapeutic taper. In my experience, the taper will be most successful if you taper more slowly both at the beginning and at the end. For example, if a patient has become dependent on taking 8 to 10 Vicodin per day, you might reduce the dose by a half pill every few days initially, speed up the taper to a full pill per unit time, and then slow it back down to a half or a quarter pill at the end.
Switching to methadone as a tapering strategy
Another option is to switch your patient to a different opiate before doing the taper. One reason to do this is to interrupt the positive associations a patient might have made with a particular drug. In addition, switching to a longer-acting drug makes for less frequent dosing. The fewer daily choices people have between taking or not taking something, the more successful they’ll be in following a tapering schedule. For example, one approach is to switch a patient from Vicodin to methadone, which has advantages because it is inexpensive, longer acting, and produces less euphoria than many of the other opioids. Not every pharmacy carries it, but many do, and it comes in a variety of forms, including liquid as well as tablets of different strengths.
Contrary to popular belief, you do not have to be part of a federally licensed treatment center to prescribe methadone. Any provider with a DEA license to prescribe Schedule II substances can prescribe methadone—as long as you are clear in your documentation that you are prescribing it for pain rather than for treatment of narcotic abuse. However, if the patient’s opiate use is not related to pain, you will need to either refer to a narcotic treatment program for methadone maintenance or to a buprenorphine provider for office-based opioid treatment.
Conversion tables are available to help you determine the right dose of methadone to use when transitioning from opioids like hydrocodone or oxycodone. For example, see the Methadone Conversion and Dosing Information table.
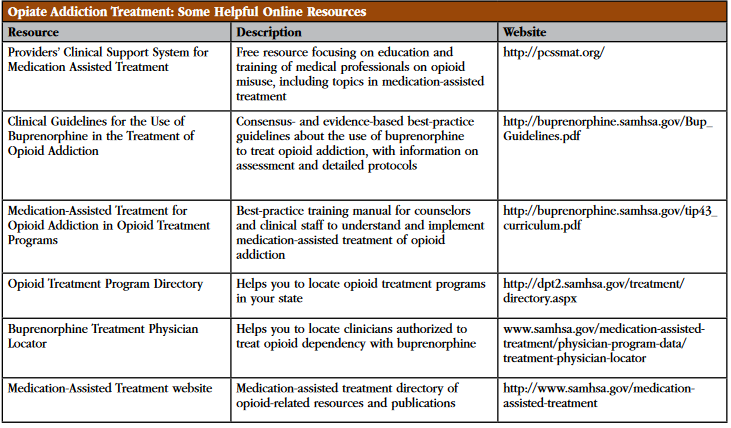
Table: Opiate Addiction Treatment: Some Helpful Online Resources
Click here to view as a full-size image .
On a cautionary note, since methadone is long-acting, patients may not notice the effects as quickly as they are used to, and might be tempted to take extra doses initially. The danger is that these doses can catch up to them all at once, and people may accidentally overdose on methadone because of their inexperience in using it.
Buprenorphine for maintenance or detox treatment
Buprenorphine is a semisynthetic opioid that has been available as a narcotic since 1981. It has long been used as an option for treating severe pain and comes in different forms, including injectable, sublingual, and as a skin patch.
How did buprenorphine end up playing such a central role in treating opiate abuse? Unlike all other opiate products such as codeine, hydrocodone, oxycodone, methadone, and even heroin, buprenorphine is an opioid agonist/antagonist. The agonist property activates opioid receptors, while the antagonist property tends to modulate that activation. For this reason, the medication helps patients feel normal rather than high.
The bottom line is that buprenorphine, with its push/pull agonist/antagonist properties, is less likely to be abused than its opiate cousins. This in itself makes it a good choice for maintenance treatment. The buprenorphine mono product (without naloxone, brand name Subutex) has been generic for a few years and is relatively cheap; some insurance companies have it on their preferred formulary list. For some addiction specialists, plain old buprenorphine is the treatment of choice for opiate maintenance or detox.
In order to be able to prescribe buprenorphine (with or without naloxone), you need a waiver from the Department of Health and Human Services—a system that was established as part of the Drug Addiction Treatment Act of 2000 (DATA2000). To obtain this waiver, you need to go through an 8-hour training, which will enable you to prescribe buprenorphine to up to 30 patients in the first year and as many as 100 patients in the second year. New federal legislation will soon allow for treatment of up to 200 patients after the second year.
Buprenorphine is a Schedule III controlled substance, so it’s less restricted than methadone (which is Schedule II). Patients can get a one-month prescription from a buprenorphine provider with up to 5 refills, although most experts recommend at least monthly visits. One of the reasons buprenorphine is less restricted is because of its safety profile—as an opioid agonist/antagonist, it’s more difficult to overdose on buprenorphine than it is on methadone.
Buprenorphine/naloxone
Given that buprenorphine alone is an effective treatment for opioid addiction, why did drug makers create Suboxone, the combination of buprenorphine and naloxone? Wouldn’t naloxone—an opiate blocker used to reverse opioid overdoses—neutralize buprenorphine’s effectiveness, rendering it useless as a treatment? Not if it’s taken sublingually, as directed. When buprenorphine is absorbed through the mouth’s mucosal lining, it works fine—but naloxone is then inactivated. But if you choose to grind the combination pill and dissolve in saline to inject it, the naloxone is very much active, and will prevent you from getting high. So naloxone was added to reduce the potential for abuse by injection.
How to start patients on buprenorphine: Induction
Induction refers to the somewhat complicated process of starting an opioid-dependent patient on buprenorphine (by “buprenorphine” I am referring to any of the preparations, whether mono or combined with naloxone). It can be done in an outpatient setting, and the process usually takes 1–2 days.
I start by emphasizing to patients that they should not use opioids for about 24 hours before presenting for induction—otherwise the first dose will cause an unpleasant withdrawal experience. For those of you who need a review, the early symptoms of opiate withdrawal are anxiety, craving, nausea, aches in the lower back areas and legs, some muscle cramps, and possibly some diarrhea. Later, it gets progressively worse to the point where patients have a runny nose, watery eyes, intestinal cramps, vomiting, muscle twitches, and the famous “cold turkey” goose flesh.
Once they’re in the early stage of withdrawal, start with low doses of buprenorphine, 2–4 mg, and then every hour or two give them additional doses until they’re comfortable. I check on patients every hour or so. We have a waiting room with wi-fi, a DVD player, and magazines, and I tell them, “You’re going to be hanging out for a while. There’s a bathroom very close by.” Medical personnel, myself or my nurse, are checking on them regularly, and they can get someone’s attention right away.
After the second dose, most people are feeling much better physically, so then it’s just a matter of titrating up to where they feel comfortable without “feeling like a zombie”—in other words, too sedated.
Depending on their tolerance, typical doses are between 12 and 16 mg a day. Some people do fine with 10 mg, while some people need more like 20 mg; it just depends on individual tolerance. If they’re feeling sleepy or a little lightheaded, stop dosing wherever they are. I’ve seen a lot of variability in dosing needs. Someone who has been using 8 or 10 Vicodin a day may get by with 8 mg of buprenorphine. But the patient who is injecting a fair amount of heroin multiple times a day may require up to 16 mg or more that first day.
At the end of the first day of induction, I make sure patients have someone who can drive them home. I will usually give them a prescription to pick up a small amount of buprenorphine at the pharmacy. If they absolutely need it, they can take a dose at home. They will rarely actually need it because buprenorphine is long-acting, and once you get the initial dose up during the course of a day, the cumulative effects will get them through at least the next 12 hours or so.
The next morning, the patients come back and we pick up where we left off the day before. I give them the first day’s dose all at once to start with and make sure they can tolerate it, and then if necessary go up from there. It usually takes us two days to find the right dose, but occasionally it takes longer.
The maximum recommended dose of buprenorphine or buprenorphine/naloxone is 32 mg per day. Due to the agonist/antagonist properties of the drug, doses higher than 32 mg may start to cause some opioid withdrawal symptoms, which is known as the “ceiling effect.” Patients who have been using high doses of opioids, either prescription opioid analgesics or heroin, may need a higher overall opioid dose than can be provided with buprenorphine due to this effect. In that case, these patients would benefit more from methadone maintenance because methadone does not have a ceiling effect. There’s no set upper limit for methadone—the final maintenance dose is based on tolerance and can be affected by other medications that enhance methadone metabolism (such as some of the antiretroviral medications to treat HIV).
After the induction, I will generally see patients weekly, which allows me to make more dosage adjustments as needed. I will increase the dose if there are still cravings and withdrawal symptoms, or reduce it if there are opioid side effects, such as constipation or oversedation.
Life after induction
Maintenance vs. tapering
Once we have arrived at a stable dose of buprenorphine, I begin talking to patients about how long they will need to stay on it. Some people need more time on the medication—many months to years—because of the severity of their addiction and the harsh consequences of relapse. Having such patients on a maintenance dose for many months allows them time to work on the behavioral components that they are learning in therapy or in 12 step programs.
On the other hand, other patients don’t have as severe a problem with opiates, and if they have good social supports, they might be ready for a taper fairly soon after they start. The taper is fairly straightforward. The smallest dosage strength is 2 mg, and people can cut pills or strips in half and taper down in 1-mg increments. I’ll have them decrease the dose anywhere from every few days to every few weeks depending on the time available and the patient’s physical and psychological comfort level. A typical outpatient taper will take around 2 months, and this time can be lengthened depending on the patient’s readiness and starting dose.
Psychosocial interventions
In addition to prescribing medication, I make sure that my patients are engaged in some kind of psychosocial intervention. This might be individual counseling, a group program, or involvement with a 12 step self-help group. I want them to be working on relapse prevention skills, coping skills, and refusal skills. And in their home environment, I encourage them to rally as much support as possible from family and friends who are not using drugs.
Psychiatric medications for other symptoms
Antidepressants and other medications for anxiety can be very helpful in supporting recovery, and just about any of the SSRIs and SNRIs are appropriate. Both gabapentin and hydroxyzine are effective anti-anxiety medications that are not controlled substances. For insomnia, there are medications like trazodone.
Benzodiazepines are not ideal for these patients. For one, they can produce a high that may lead to craving and relapse to opiates. In addition, it’s hard to overdose on buprenorphine alone, the combination of buprenorphine and benzodiazepines can more readily lead to overdose. It’s important to let patients know about this risk.
Typical causes of failure of buprenorphine treatment
Lack of support
Lack of good support in the immediate home environment is a big reason why patients may have significant problems remaining abstinent. They need to have family or close non-using friends for encouragement and to help them deal with the emotional and physical ups and downs of being in early recovery.
Using other drugs
Some people don’t want to give up their marijuana or their alcohol, which is another reason for relapse back to opioids. I do random drug testing, and I let people know that the goal is going to be abstinence from all the substances that could cause problems.
Even though maintenance methadone or buprenorphine only target withdrawal from and cravings for opioids as a class, most people will also reduce their intake of other drugs of abuse. This is because the behavioral components of the recovery program are universal, not specific to opioids.
How to choose among the different formulations of buprenorphine
There are several different formulations of buprenorphine available (see our table, “Agents Used in Medication-Assisted Treatment (MAT) for Opioid Addiction"). Suboxone is the most common combination product and the only one that is FDA approved for induction. It was originally available in sublingual tablets, and now it’s available as strips or films, which are also sublingual. There’s also a different brand name of tablet (Zubsolv), which is a more bioavailable form of buprenorphine. Higher bioavailability means that a larger fraction of the drug is absorbed, which in turn allows for a lower number of milligrams to achieve the same effect. A more recent alternative is Bunavail, a buccal film that is also highly bioavailable and which dissolves on the inside of the cheek as opposed to under the tongue.
The main factor influencing the choice of formulation tends to be what the patient’s insurance policy covers. Some companies simply cover the cheapest product—which is the generic mono product, buprenorphine without naloxone. Other companies will preferentially cover the combination product, typically the Suboxone strips.
In general, I favor the combination product over the mono product, in part because it is more available in pharmacies. Some patients and their families request the combination because they believe having the naloxone adds a measure of safety, although in my experience the scenario the naloxone is meant to prevent (injection abuse) is rarely a concern. The mono product should always be used, however, for pregnant women in order to reduce exposure to medications overall, including naloxone.
Among the combination formulations, Suboxone has been around the longest and is most familiar to patients. The strips tend to be more easily dissolvable than the former Suboxone tablets, which are no longer marketed. It takes about 5 minutes to dissolve under the tongue, and during that time, the patient shouldn’t talk or swallow (not a fun way to start the day).
The mono buprenorphine tablets take at least 5 minutes to dissolve, and it can take longer if you need two or more tablets. I tell my patients to rinse their mouth out really thoroughly, and then to get it nice and moist before putting the tablet under the tongue, because that will help everything to dissolve.
The Bunavail product is the most recent one to come to market. The claim is that it dissolves the fastest and allows you to talk and eat with the patch on the inside of your cheek.
For each strength of Suboxone film, there is an equivalent strength Zubsolv tablet or Bunavail buccal film, but the equivalent strength does not mean the same number of milligrams of buprenorphine/naloxone due to differences in bioavailability between the different products. The conversion charts tell practitioners what dose of Zubsolv or Bunavail to use for a known dose of Suboxone being taken by your patient. That helps to reduce confusion.
Vivitrol
Vivitrol is a long-acting injectable form of naltrexone, which like naloxone is an opiate receptor blocker. Before an injection with Vivitrol, people have to abstain from opioids for at least several days, preferably about one week. You may have to do a detox with methadone or buprenorphine to get patients off their preferred opiates, and then have them stop for a few days. The injections are every 4 weeks, and they can’t be done at home. They are deep gluteal intramuscular injections, and the manufacturer sends kits to the physician’s office. You don’t need any special licensing to administer it; any psychiatrist or primary care practitioner can do it in the office.
Vivitrol is certainly effective to extinguish opioid use, but the potential problem is that people may not show up for subsequent injections. In a psychiatric setting, giving someone who has a dual diagnosis an injection can give both the provider and the patient some peace of mind for a while, so that can be a part of discharge planning.
Conclusion
While the opiate epidemic is serious, we have some excellent tools for helping patients decrease their use of the more dangerous drugs, and in some cases patients can successfully end their opiate use completely. According to the latest follow-up studies, buprenorphine retention rates at 1 year are around 60%–66% (Fiellin et al, N Engl J Med 2006;355:365–374). As far as methadone maintenance, those rates are as high as 80%, according to one recent study (Hser YI et al, Addiction 2014;109(1):79–87). Medication-assisted treatment for opiate addiction works and should be part of our therapeutic repertoire.
Table: Risk Factors For Opioid Overdose
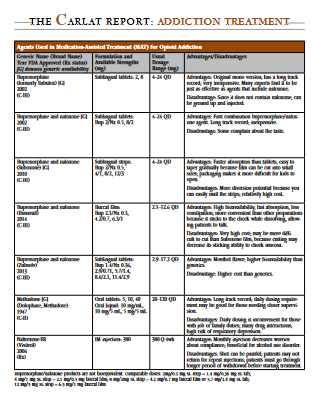
Table: Agents Used in Medication-Assisted Treatment (MAT) for Opioid Addiction

Newsletters
Please see our Terms and Conditions, Privacy Policy, Subscription Agreement, Use of Cookies, and Hardware/Software Requirements to view our website.
© 2026 Carlat Publishing, LLC and Affiliates, All Rights Reserved.


_-The-Breakthrough-Antipsychotic-That-Could-Change-Everything.webp?t=1729528747)



