Home » What’s New with the FDA Labeling for Pregnancy and Lactation?
What’s New with the FDA Labeling for Pregnancy and Lactation?
July 1, 2016
From The Carlat Psychiatry Report
 Talia Puzantian, PharmD, BCPP
Deputy Editor, The Carlat Psychiatry Report
Dr. Puzantian has disclosed that she has no relevant financial or other interests in any commercial companies pertaining to this educational activity.
Talia Puzantian, PharmD, BCPP
Deputy Editor, The Carlat Psychiatry Report
Dr. Puzantian has disclosed that she has no relevant financial or other interests in any commercial companies pertaining to this educational activity.
We’ve been hearing for some time now about the FDA’s plans to revise the long-standing categories A, B, C, D, and X designations for risk of using medications in pregnancy. The new rule (referred to as PLLR for Pregnancy and Lactation Labeling Rule) was proposed in 2008, finalized in 2014, and implementation began during the summer of 2015.
What was wrong with the old system?
The old pregnancy categories appeared, at least superficially, to be pretty useful. If a drug was labeled “A” it was considered completely safe (only 0.7% of medications in U.S. were considered “A”), “B” was pretty safe, “C” was possibly harmful (most medications fell into this category), “D” was probably harmful but still usable if benefits outweighed risks, and finally “X” was positively teratogenic and not to be used ever in pregnancy. In addition to the grades, companies supplied brief descriptions of the risks, but were not required to describe the data in any systematic way.
There were a few problems with the old system. First, the categories were overly simplistic and couldn’t adequately describe the potential for negative outcomes. Many would understandably assume that two medications in the same category must have the same level of risk, but this was not true. For example, both lithium and valproate had been classified as category D, yet the risk of cardiac malformation with lithium is roughly 0.05%–0.1% compared to the 8% risk for neural tube defects with valproate. Complicating things even further for women and their providers was the FDA’s ruling that valproate would be considered category X for migraines but would remain category D for other indications.
Another critical issue was that categorization didn’t differentiate between information based on animal studies and information gleaned from human studies. For example, new medications could earn a benign category B classification even if there was not a single human study showing safety—as long as animal studies showed no fetal risk. And when new data did become available, changes in categorization were not consistent—yet another problem. You might remember that paroxetine (Paxil) was a category C drug, but when several studies suggested a higher risk for cardiac malformations in exposed fetuses, the FDA changed its status to category D. Later, other studies didn’t find this increased risk, but the category designation remained the same (see expert interview with Dr. Burt for more on this issue). The old system allowed a newer medication to be considered innocent until proven guilty, but an older medication with decades of safe experience in women would be considered category C just because of some fetal risk shown in animal studies. This was misleading to clinicians. In the end, while the old system was quick and dirty, it turns out it wasn’t very clinically meaningful.
How is the new system different?
1. Sections are shuffled
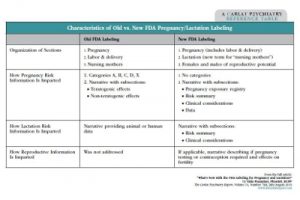
The old labeling included three sections: pregnancy, labor & delivery, and nursing mothers. In the new system, “pregnancy” includes both pregnancy and labor & delivery, the old term “nursing mothers” is replaced with “lactation,” and a new, third section is created, called “females and males of reproductive potential.” This last section includes information about pregnancy testing, contraception, and effects on fertility.
2. Instead of letter grades, a longer narrative
The new system abolishes letter grades for drugs and instead requires that manufacturers provide narrative descriptions of risks. The information is organized into three subsections: risk summary, clinical considerations, and data. In addition, if a pregnancy exposure registry exists, you will be given the website or other ways to access it. The idea is that you will be able to read about the risks and then draw your own conclusions about whether the risk/benefit ratio justifies prescribing a drug.
How well does this work in practice? Let’s take an example of a recently approved psychiatric drug, cariprazine (Vraylar). You can access the package insert here. Now compare cariprazine’s labeling with the old labeling for another relatively new antipsychotic, lurasidone (Latuda) here.
Looking at these two package inserts, you’ll see that the new system (used for cariprazine, which was approved in 2015) provides much more detailed information and has no letter grade. The lurasidone label gives us a reassuring B grade, but explains this grade with uninformative statements, ie, that we have no data in humans, either in pregnancy or breastfeeding. With cariprazine, we also have only animal data presented, but at least the label provides contact information for the registry, which over time will be populated with human data. The cariprazine label also adds useful information about the background risk for malformation and the drug class risks.
Since cariprazine and lurasidone are still relatively new drugs, their current data are limited, but eventually the narrative will include a wealth of details, such as risks associated with underlying illness, necessary dosage adjustments in pregnancy, and effects on labor and delivery.
The old letter grade had the benefit of providing a quick and dirty summary, but as we are learning more about drugs in pregnancy, such a shorthand is no longer feasible. That’s unfortunate, because it means we have to slog through narratives that will become longer and longer over time. Luckily, there are publications such as TCPR and others whose mission in life is to synthesize this data for you.
Another component of the new PLLR is a statement about background risk of adverse pregnancy outcomes: eg, “The estimated background risk of major birth defects and miscarriage for the indicated populations is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2%–4% and 15%–20%, respectively.”
When does the new system actually begin?
The new system is already in place for all medications approved after June of 2015. For meds that were approved between 2001 and 2015, companies have until 2020 to create the revisions. Finally, for meds approved before 2001, don’t hold your breath—the FDA recommends, but does not require, manufacturers to provide new data for these older medications.
TCPR Verdict: The new labeling has received rave reviews from heavyweight organizations such as The American Academy of Pediatrics and the American College of Obstetrics and Gynecology. Overall, we think this is a step in the right direction, but the longer narratives will be challenging for busy prescribers.
General PsychiatryWhat was wrong with the old system?
The old pregnancy categories appeared, at least superficially, to be pretty useful. If a drug was labeled “A” it was considered completely safe (only 0.7% of medications in U.S. were considered “A”), “B” was pretty safe, “C” was possibly harmful (most medications fell into this category), “D” was probably harmful but still usable if benefits outweighed risks, and finally “X” was positively teratogenic and not to be used ever in pregnancy. In addition to the grades, companies supplied brief descriptions of the risks, but were not required to describe the data in any systematic way.
There were a few problems with the old system. First, the categories were overly simplistic and couldn’t adequately describe the potential for negative outcomes. Many would understandably assume that two medications in the same category must have the same level of risk, but this was not true. For example, both lithium and valproate had been classified as category D, yet the risk of cardiac malformation with lithium is roughly 0.05%–0.1% compared to the 8% risk for neural tube defects with valproate. Complicating things even further for women and their providers was the FDA’s ruling that valproate would be considered category X for migraines but would remain category D for other indications.
Another critical issue was that categorization didn’t differentiate between information based on animal studies and information gleaned from human studies. For example, new medications could earn a benign category B classification even if there was not a single human study showing safety—as long as animal studies showed no fetal risk. And when new data did become available, changes in categorization were not consistent—yet another problem. You might remember that paroxetine (Paxil) was a category C drug, but when several studies suggested a higher risk for cardiac malformations in exposed fetuses, the FDA changed its status to category D. Later, other studies didn’t find this increased risk, but the category designation remained the same (see expert interview with Dr. Burt for more on this issue). The old system allowed a newer medication to be considered innocent until proven guilty, but an older medication with decades of safe experience in women would be considered category C just because of some fetal risk shown in animal studies. This was misleading to clinicians. In the end, while the old system was quick and dirty, it turns out it wasn’t very clinically meaningful.
How is the new system different?
1. Sections are shuffled
The old labeling included three sections: pregnancy, labor & delivery, and nursing mothers. In the new system, “pregnancy” includes both pregnancy and labor & delivery, the old term “nursing mothers” is replaced with “lactation,” and a new, third section is created, called “females and males of reproductive potential.” This last section includes information about pregnancy testing, contraception, and effects on fertility.
2. Instead of letter grades, a longer narrative
The new system abolishes letter grades for drugs and instead requires that manufacturers provide narrative descriptions of risks. The information is organized into three subsections: risk summary, clinical considerations, and data. In addition, if a pregnancy exposure registry exists, you will be given the website or other ways to access it. The idea is that you will be able to read about the risks and then draw your own conclusions about whether the risk/benefit ratio justifies prescribing a drug.
How well does this work in practice? Let’s take an example of a recently approved psychiatric drug, cariprazine (Vraylar). You can access the package insert here. Now compare cariprazine’s labeling with the old labeling for another relatively new antipsychotic, lurasidone (Latuda) here.
Looking at these two package inserts, you’ll see that the new system (used for cariprazine, which was approved in 2015) provides much more detailed information and has no letter grade. The lurasidone label gives us a reassuring B grade, but explains this grade with uninformative statements, ie, that we have no data in humans, either in pregnancy or breastfeeding. With cariprazine, we also have only animal data presented, but at least the label provides contact information for the registry, which over time will be populated with human data. The cariprazine label also adds useful information about the background risk for malformation and the drug class risks.
Since cariprazine and lurasidone are still relatively new drugs, their current data are limited, but eventually the narrative will include a wealth of details, such as risks associated with underlying illness, necessary dosage adjustments in pregnancy, and effects on labor and delivery.
The old letter grade had the benefit of providing a quick and dirty summary, but as we are learning more about drugs in pregnancy, such a shorthand is no longer feasible. That’s unfortunate, because it means we have to slog through narratives that will become longer and longer over time. Luckily, there are publications such as TCPR and others whose mission in life is to synthesize this data for you.
Another component of the new PLLR is a statement about background risk of adverse pregnancy outcomes: eg, “The estimated background risk of major birth defects and miscarriage for the indicated populations is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2%–4% and 15%–20%, respectively.”
Table: Characteristics of Old vs. New FDA Pregnancy/Lactation Labeling
(Click to view full-size PDF.)
When does the new system actually begin?
The new system is already in place for all medications approved after June of 2015. For meds that were approved between 2001 and 2015, companies have until 2020 to create the revisions. Finally, for meds approved before 2001, don’t hold your breath—the FDA recommends, but does not require, manufacturers to provide new data for these older medications.
TCPR Verdict: The new labeling has received rave reviews from heavyweight organizations such as The American Academy of Pediatrics and the American College of Obstetrics and Gynecology. Overall, we think this is a step in the right direction, but the longer narratives will be challenging for busy prescribers.
KEYWORDS pharmaceutical_industry practice-tools-and-tips psychopharmacology_tips women's_issues_in_psychiatry

Issue Date: July 1, 2016
Table Of Contents
Recommended
Newsletters
Please see our Terms and Conditions, Privacy Policy, Subscription Agreement, Use of Cookies, and Hardware/Software Requirements to view our website.
© 2026 Carlat Publishing, LLC and Affiliates, All Rights Reserved.


_-The-Breakthrough-Antipsychotic-That-Could-Change-Everything.webp?t=1729528747)



