Home » Using Psychiatric Biomarkers in your Practice
Using Psychiatric Biomarkers in your Practice
May 1, 2015
From The Carlat Psychiatry Report
Steve Balt, MD, MS
Board Member, The Carlat Report. In private practice in the San Francisco Bay area.
Dr. Balt discloses that his spouse is employed as a sales representative for Otsuka America.
Treatment of the psychiatric patient is as much an art as it is a science. Diagnosing and deciding on treatments in psychiatry is based on a series of human interactions. We observe and we listen to patients, we ask questions, we consider their responses, and we synthesize all this information in order to render a judgment. Such judgments are subjective. We think this patient has bipolar disorder, but could it be schizoaffective? We think lithium will work best, but perhaps we should start with lurasidone? Choosing the “right” treatment for a given patient involves a large number of factors, including personal preference—both the clinician’s and the patient’s.
Is there a better way? Of course there is—most other medical specialists can order labs that provide objective evidence used to guide decisions. These are called “biomarkers.” Psychiatrists have been searching for biomarkers for quite a while. Some of you will remember the dexamethasone suppression test (DST) from the 1960s—which was billed as a biomarker for depression. The biological hypothesis was that people with depression had excessive production of the stress hormone cortisol. Normally, giving healthy people an injection of the steroid dexamethasone would suppress cortisol production. But if depressed patients were churning out too much cortisol, dexamethasone would not suppress cortisol as much as it should—leading to a “positive” DST. Unfortunately, after years of research, the test didn’t pan out, because it had a low sensitivity, 49-67% by some reports, and as low as 14% in depressed outpatients (Health and Public Policy Committee, ACP, Ann Int Med 1984;100:307–308).
Recently, we’ve seen a spate of potential biomarkers for various psychiatric disorders. Before examining the evidence, there is a major inherent limitation to all of this research, which is that we have no true gold standard diagnostic test in psychiatry against which we can judge the accuracy of a new test. Other specialties have such gold standards (such as biopsies for cancer or cardiac catheterization for coronary artery disease). At this point, our only gold standard is the interview.
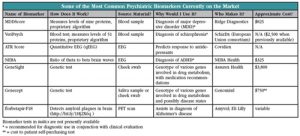
Recently introduced biomarkers in psychiatry come in three flavors. They can be diagnostic (does the patient have a disease?); prognostic (will the patient develop a disease?); or predictive or “theranostic” (will the patient respond to a particular therapy?) (Weickert CS, et al, Disease Markers 2013;35(1):3-9). While we don’t have room to review each and every potential biomarker out there, we will focus on some that have received the most attention. See the chart below for a list of all biomarkers being marketed (or close to being marketed) in psychiatry.
Diagnostic Biomarkers
MDDScore. The MDDScore, marketed by Ridge Diagnostics, is a blood test that purports to diagnose depression. It measures the levels of nine proteins in a single sample of the patient’s blood. These values are fed into an algorithm and a score is reported, ranging from one to nine, where anything greater than or equal to five represents “90% likelihood of depression,” according to their patient materials.
Two studies have evaluated the accuracy of the test. In the first study, blood samples were taken from 70 depressed patients and 43 non-depressed patients in a clinical population, and the algorithm predicted a diagnosis of depression with 91% sensitivity and 81% specificity (Papakostas GI, Shelton RC et al, Molec Psychiat 2011;18:332–339). This sounds impressive, but all we can really say is that the depressed patients differed from healthy controls on certain measures of inflammation, stress response, and metabolism. We don’t know whether patients with other conditions such as bipolar disorder or schizophrenia share the same profile, nor do we know the formula. Notably, the study excluded patients on antidepressants, antipsychotics, or NSAIDs in the two weeks prior to the blood test, and those patients with “serious and/or unstable medical disorders.” (For more, see the chart Sensitivity and Specificity: A Refresher below.)
A follow-up study was published earlier this year, which also demonstrated impressive sensitivity (96%) and specificity (86%) on a similarly selected group of 68 depressed and 86 non-depressed patients. However, while they measured the same nine proteins, they didn’t use the same algorithm as in the earlier study. In fact, they “trained” the algorithm—a fancy way of saying they ran it over and over on a subgroup (N=102) of the total subjects, tweaking the algorithm each time to make it positive for the depressed patients and negative for the non-depressed patients, and then “validated” it on the remaining 52 patients. They also made adjustments for gender and body mass index (BMI) and normalized cortisol levels, which the initial study did not do.
In other words, the “MDDScore” seems to measure something of interest in patients with depression, but we don’t know how—or whether—it differentiates depression from other disorders, nor whether it can guide treatment (eg, which SSRI or SNRI, or maybe psychotherapy). And with the two replication studies published by the manufacturer, both with proprietary algorithms, it’s hard to know what’s exactly being tested when you order this test. At a cost of $826.45 ($385 if the patient pays within 30 days), but with coverage by “most insurance companies,” according to Ridge Diagnostics, we’re not so sure how this test adds any information that improves upon treatment as usual.
VeriPsych. The VeriPsych assay, introduced in 2010, measures the levels of 51 proteins in a single blood sample with the goal of diagnosing schizophrenia. Selection of the 51 proteins was similar to the process used in the MDDScore, with a few differences. Basically, researchers measured the levels of 181 proteins from serum samples of 250 acutely psychotic, treatment-naïve patients and 230 controls. They found significant differences in the levels of 22 of these proteins. Nine additional proteins were added to the assay because they have been “associated with schizophrenia” in earlier research, while an additional 20 proteins were selected because they’re expressed differently in patients with bipolar disorder; they included these proteins in hopes of differentiating between the two disorders.
As with the MDDScore, they developed a mathematical “decision rule” based on levels of these proteins in a test sample of 577 schizophrenic patients and 229 controls. The resulting formula correctly identified schizophrenia in this population with a sensitivity and specificity of 83% each (Schwarz E, Izmailov R et al, Biomarker Insights 2010;5:39–47).
The VeriPsych test was initially offered at a cost of $2,500. In a 2011 interview, the Chief Medical Officer of Myriad RBM, developer of the test, reported that “hundreds of patients” had been tested and that it was covered by “numerous insurance carriers” (Kaplan A. Blood Tests for Diagnosis of Schizophrenia and Depression Psychiatr Times. 2011;28(8)). However, in January 2013 the company suspended the test. The high cost may have been one factor, as well as the fact that the test failed to differentiate between schizophrenia and other psychiatric conditions (source: http://bit.ly/1eWorX2). Meanwhile, many of the same scientists have moved to the SchizDx project (http://schizdx.pera.com/) to continue work on the test, so stay tuned.
Predictive Biomarkers
ATR Score. The ATR score is based on using quantitative EEG (qEEG) to predict response to particular antidepressants (See TCPR, November 2009 for earlier coverage of the test). The score incorporates three frontal EEG features (theta power, alpha power, and alpha2 power) measured at baseline and one week after a patient starts medication. The scores are given on a scale of 0 to 100, which corresponds to probability of response.
Early studies showed that the ATR score predicted response to escitalopram (Lexapro) with an accuracy of 74%, while other measures like serum drug levels and genetic polymorphisms were not predictive (Leuchter AF, Cook IA et al, Psychiatry Res 2009;169:124–131). In a separate study, the ATR score showed a different result in responders to fluoxetine than in those who responded to placebo, indicating a treatment-specific biological change (Hunter AM et al, J Clin Neurophysiol 2011;28(5):478–482).
Despite years of research and having teamed up with a medical device company (Aspect Medical, now a division of Covidien) the ATR has not yet reached clinical practice. If and when it does, the value of enhanced (though not perfect) treatment selection must be balanced against the significant capital investment and technical expertise it will undoubtedly require. So its true utility may be limited.
Others. Not a month goes by without a new biomarker study published in a major psychiatric journal. But most need to be replicated and, as of yet, none can be easily ordered from your office. For instance, a study showed last year that reduced expression and increased methylation of a gene called SKA2 was linked to suicidal behavior (Guintivano J et al, Am J Psychiatry 2014;171(12):1287–1296). Another group found that suicidal ideation was associated with differential expression of six RNA transcripts in peripheral blood (SKA2 was not among those that made the cut) (Le-Niculescu H et al, Mol Psychiatry 2013;18(12):1249–1264).
Brain imaging can also hint at new biomarkers. Researchers at Emory University have used PET scans to show that low activity of the right anterior insular cortex predicts a good response to psychotherapy, while high activity predicts a response to escitalopram (McGrath CL et al, JAMA Psychiatry 2013;70(8):821–829). And the list goes on and on.
The “Holy Grail” of Psychiatry?
While diagnostic biomarkers might someday revolutionize psychiatric diagnosis that day is not yet here. The experts still emphasize the value of the clinical interview and advise the use of biomarker tests as confirmatory or supportive evidence. However, one potential advantage of biomarkers is to identify new targets for medications and other physiological therapies, as in other areas of “precision medicine.”
The operative word here is “might.” Despite decades of research dating back to the DST, no biomarker test has revolutionized psychiatric care. Even the most commonly used biomarkers—pharmacogenetics—are questionable in their ability to improve psychiatric care over treatment as usual (see the companion article on GeneSight in this issue).
General PsychiatryIs there a better way? Of course there is—most other medical specialists can order labs that provide objective evidence used to guide decisions. These are called “biomarkers.” Psychiatrists have been searching for biomarkers for quite a while. Some of you will remember the dexamethasone suppression test (DST) from the 1960s—which was billed as a biomarker for depression. The biological hypothesis was that people with depression had excessive production of the stress hormone cortisol. Normally, giving healthy people an injection of the steroid dexamethasone would suppress cortisol production. But if depressed patients were churning out too much cortisol, dexamethasone would not suppress cortisol as much as it should—leading to a “positive” DST. Unfortunately, after years of research, the test didn’t pan out, because it had a low sensitivity, 49-67% by some reports, and as low as 14% in depressed outpatients (Health and Public Policy Committee, ACP, Ann Int Med 1984;100:307–308).
Recently, we’ve seen a spate of potential biomarkers for various psychiatric disorders. Before examining the evidence, there is a major inherent limitation to all of this research, which is that we have no true gold standard diagnostic test in psychiatry against which we can judge the accuracy of a new test. Other specialties have such gold standards (such as biopsies for cancer or cardiac catheterization for coronary artery disease). At this point, our only gold standard is the interview.
Recently introduced biomarkers in psychiatry come in three flavors. They can be diagnostic (does the patient have a disease?); prognostic (will the patient develop a disease?); or predictive or “theranostic” (will the patient respond to a particular therapy?) (Weickert CS, et al, Disease Markers 2013;35(1):3-9). While we don’t have room to review each and every potential biomarker out there, we will focus on some that have received the most attention. See the chart below for a list of all biomarkers being marketed (or close to being marketed) in psychiatry.
Diagnostic Biomarkers
MDDScore. The MDDScore, marketed by Ridge Diagnostics, is a blood test that purports to diagnose depression. It measures the levels of nine proteins in a single sample of the patient’s blood. These values are fed into an algorithm and a score is reported, ranging from one to nine, where anything greater than or equal to five represents “90% likelihood of depression,” according to their patient materials.
Two studies have evaluated the accuracy of the test. In the first study, blood samples were taken from 70 depressed patients and 43 non-depressed patients in a clinical population, and the algorithm predicted a diagnosis of depression with 91% sensitivity and 81% specificity (Papakostas GI, Shelton RC et al, Molec Psychiat 2011;18:332–339). This sounds impressive, but all we can really say is that the depressed patients differed from healthy controls on certain measures of inflammation, stress response, and metabolism. We don’t know whether patients with other conditions such as bipolar disorder or schizophrenia share the same profile, nor do we know the formula. Notably, the study excluded patients on antidepressants, antipsychotics, or NSAIDs in the two weeks prior to the blood test, and those patients with “serious and/or unstable medical disorders.” (For more, see the chart Sensitivity and Specificity: A Refresher below.)
A follow-up study was published earlier this year, which also demonstrated impressive sensitivity (96%) and specificity (86%) on a similarly selected group of 68 depressed and 86 non-depressed patients. However, while they measured the same nine proteins, they didn’t use the same algorithm as in the earlier study. In fact, they “trained” the algorithm—a fancy way of saying they ran it over and over on a subgroup (N=102) of the total subjects, tweaking the algorithm each time to make it positive for the depressed patients and negative for the non-depressed patients, and then “validated” it on the remaining 52 patients. They also made adjustments for gender and body mass index (BMI) and normalized cortisol levels, which the initial study did not do.
In other words, the “MDDScore” seems to measure something of interest in patients with depression, but we don’t know how—or whether—it differentiates depression from other disorders, nor whether it can guide treatment (eg, which SSRI or SNRI, or maybe psychotherapy). And with the two replication studies published by the manufacturer, both with proprietary algorithms, it’s hard to know what’s exactly being tested when you order this test. At a cost of $826.45 ($385 if the patient pays within 30 days), but with coverage by “most insurance companies,” according to Ridge Diagnostics, we’re not so sure how this test adds any information that improves upon treatment as usual.
VeriPsych. The VeriPsych assay, introduced in 2010, measures the levels of 51 proteins in a single blood sample with the goal of diagnosing schizophrenia. Selection of the 51 proteins was similar to the process used in the MDDScore, with a few differences. Basically, researchers measured the levels of 181 proteins from serum samples of 250 acutely psychotic, treatment-naïve patients and 230 controls. They found significant differences in the levels of 22 of these proteins. Nine additional proteins were added to the assay because they have been “associated with schizophrenia” in earlier research, while an additional 20 proteins were selected because they’re expressed differently in patients with bipolar disorder; they included these proteins in hopes of differentiating between the two disorders.
As with the MDDScore, they developed a mathematical “decision rule” based on levels of these proteins in a test sample of 577 schizophrenic patients and 229 controls. The resulting formula correctly identified schizophrenia in this population with a sensitivity and specificity of 83% each (Schwarz E, Izmailov R et al, Biomarker Insights 2010;5:39–47).
The VeriPsych test was initially offered at a cost of $2,500. In a 2011 interview, the Chief Medical Officer of Myriad RBM, developer of the test, reported that “hundreds of patients” had been tested and that it was covered by “numerous insurance carriers” (Kaplan A. Blood Tests for Diagnosis of Schizophrenia and Depression Psychiatr Times. 2011;28(8)). However, in January 2013 the company suspended the test. The high cost may have been one factor, as well as the fact that the test failed to differentiate between schizophrenia and other psychiatric conditions (source: http://bit.ly/1eWorX2). Meanwhile, many of the same scientists have moved to the SchizDx project (http://schizdx.pera.com/) to continue work on the test, so stay tuned.
Predictive Biomarkers
ATR Score. The ATR score is based on using quantitative EEG (qEEG) to predict response to particular antidepressants (See TCPR, November 2009 for earlier coverage of the test). The score incorporates three frontal EEG features (theta power, alpha power, and alpha2 power) measured at baseline and one week after a patient starts medication. The scores are given on a scale of 0 to 100, which corresponds to probability of response.
Early studies showed that the ATR score predicted response to escitalopram (Lexapro) with an accuracy of 74%, while other measures like serum drug levels and genetic polymorphisms were not predictive (Leuchter AF, Cook IA et al, Psychiatry Res 2009;169:124–131). In a separate study, the ATR score showed a different result in responders to fluoxetine than in those who responded to placebo, indicating a treatment-specific biological change (Hunter AM et al, J Clin Neurophysiol 2011;28(5):478–482).
Despite years of research and having teamed up with a medical device company (Aspect Medical, now a division of Covidien) the ATR has not yet reached clinical practice. If and when it does, the value of enhanced (though not perfect) treatment selection must be balanced against the significant capital investment and technical expertise it will undoubtedly require. So its true utility may be limited.
Others. Not a month goes by without a new biomarker study published in a major psychiatric journal. But most need to be replicated and, as of yet, none can be easily ordered from your office. For instance, a study showed last year that reduced expression and increased methylation of a gene called SKA2 was linked to suicidal behavior (Guintivano J et al, Am J Psychiatry 2014;171(12):1287–1296). Another group found that suicidal ideation was associated with differential expression of six RNA transcripts in peripheral blood (SKA2 was not among those that made the cut) (Le-Niculescu H et al, Mol Psychiatry 2013;18(12):1249–1264).
Brain imaging can also hint at new biomarkers. Researchers at Emory University have used PET scans to show that low activity of the right anterior insular cortex predicts a good response to psychotherapy, while high activity predicts a response to escitalopram (McGrath CL et al, JAMA Psychiatry 2013;70(8):821–829). And the list goes on and on.
The “Holy Grail” of Psychiatry?
While diagnostic biomarkers might someday revolutionize psychiatric diagnosis that day is not yet here. The experts still emphasize the value of the clinical interview and advise the use of biomarker tests as confirmatory or supportive evidence. However, one potential advantage of biomarkers is to identify new targets for medications and other physiological therapies, as in other areas of “precision medicine.”
The operative word here is “might.” Despite decades of research dating back to the DST, no biomarker test has revolutionized psychiatric care. Even the most commonly used biomarkers—pharmacogenetics—are questionable in their ability to improve psychiatric care over treatment as usual (see the companion article on GeneSight in this issue).
KEYWORDS addiction diagnostic_testing genetics_and_psychiatry laboratory_testing_in_psychiatry practice-tools-and-tips
Issue Date: October 3, 2015
Table Of Contents
Recommended
Newsletters
Please see our Terms and Conditions, Privacy Policy, Subscription Agreement, Use of Cookies, and Hardware/Software Requirements to view our website.
© 2026 Carlat Publishing, LLC and Affiliates, All Rights Reserved.



_-The-Breakthrough-Antipsychotic-That-Could-Change-Everything.webp?t=1729528747)



