Home » Pharmacotherapy for Stimulant Use Disorders
Pharmacotherapy for Stimulant Use Disorders
May 21, 2021
From The Carlat Addiction Treatment Report
Rehan Aziz, MD.
Associate Professor of Psychiatry and Neurology, Rutgers Robert Wood Johnson Medical School.
Dr. Aziz has disclosed no relevant financial or other interests in any commercial companies pertaining to this educational activity.
Stimulant use is surprisingly common. In 2017, 5.5 million people in the US took cocaine, and 757,000 of these people used crack. Another 5.1 million people misused prescription stimulants, representing nearly 2% of the total population (SAMHSA, 2019; www.samhsa.gov/data/). Many pharmacological trials for stimulant use disorder have been conducted, but they have yielded mixed results at best, and there are no FDA-approved agents for stimulant use disorder. However, some medications have shown promise, particularly in conjunction with psychosocial interventions, and we’ll review them in this article.
Medications
Antidepressants
Antidepressants are thought to work in stimulant use disorder by treating stimulant-induced depression and withdrawal dysphoria. A Cochrane systematic review looked at the evidence in cocaine use disorder (CUD) by appraising 37 randomized controlled trials with 3,551 total participants. Studies included an array of antidepressants, most commonly bupropion, desipramine, and fluoxetine. Depression improved, but there was no reduction in cocaine use (Pani PP et al, Cochrane Database Syst Rev 2011(12):CD002950).
Although antidepressants are not particularly effective as a class in the management of stimulant use disorder, there is some evidence for mirtazapine in methamphetamine use disorder. Two placebo-controlled trials, both conducted in a population of men who have sex with men, demonstrated that 30 mg nightly dosing of mirtazapine modestly decreased methamphetamine use. The first study, which was relatively small at 56 participants, showed a decrease in methamphetamine-positive urine tox screens (p = 0.04) over 12 weeks when coupled with regular counseling (Colfax GN et al, Arch Gen Psych 2011;68(11):1168–1175). A larger, more recent study (n = 120) also showed a significant decrease in methamphetamine use that persisted out to 36 weeks (p = 0.02). Adherence in both studies was poor, however, and could present a challenge in the real world.
There might be new hope for bupropion in the treatment of methamphetamine use disorder as well. Recently, a large 12-week double-blind, placebo-controlled trial tested the combination of bupropion and naltrexone (Trivedi MH et al, N Engl J Med 2021;384(2):140–153). 628 adults received either extended-release injectable naltrexone (380 mg every 3 weeks) + oral extended-release bupropion (450 mg daily) or placebo. Patients who received the active drugs had fewer urine tox screens positive for methamphetamine than patients who received placebo, though the total number of negative tox screens was quite low in both groups (13.6% vs 2.5%). Mirtazapine and bupropion combined with naltrexone could be worth keeping in your back pocket. Give mirtazapine particular consideration if your patient struggles with insomnia or weight loss, which are both common in those who misuse stimulants.
Cocaine vaccine
Believe it or not, researchers have been developing a cocaine vaccine for years. The vaccine is supposed to work by producing cocaine-specific antibodies that block cocaine’s passage into the brain, thereby preventing the reinforcing effects of drug use. Although an intriguing idea, results were largely negative in two large trials (Martell BA et al, Arch Gen Psychiatry 2009;66(10):1116–1123; Kosten TR et al, Drug Alcohol Depend 2014;140:42–47). Research continues, but as of now, the cocaine vaccine is not ready for clinical use.
Disulfiram
Disulfiram isn’t just for alcohol use disorder (AUD) anymore—it holds promise for CUD, too. Recall that disulfiram causes an unpleasant reaction in patients who consume alcohol through its inhibition of acetaldehyde dehydrogenase. It also increases dopamine levels by blocking dopamine beta-hydroxylase. This could work to counter the dopamine depletion seen in chronic cocaine use and reduce cravings. Disulfiram also inhibits enzymes that metabolize cocaine. This leads to increased cocaine plasma levels, making cocaine use aversive.
Early studies with disulfiram were promising. However, it performed poorly in more recent studies, separating from placebo for length of cocaine abstinence and reduction in cocaine use in only one out of four trials. The studies, taken together, establish low-quality evidence to support the use of disulfiram (Ronsley C et al, PLoS ONE 2020;15(6):e0234809). You might keep it in mind, though, for patients who have failed other medications or for those with comorbid AUD.
GABAergic medications
GABA is the major inhibitory neurotransmitter of the CNS. By blocking activity of the dopamine reward system, GABAergic agents could prevent relapses by curbing stimulant-induced euphoria and reducing cravings. At least, that’s the theory.
Topiramate is the best studied and has yielded mixed results; out of five studies; only two were positive. Those two found that at doses of 200 mg and 300 mg daily, topiramate promoted abstinence in cocaine, amphetamine, and methamphetamine use disorders (Ronsley et al, 2020). Like disulfiram, topiramate may be especially helpful in patients with comorbid AUD.
Modafinil
The theoretical underpinning of using modafinil for stimulant use disorder is appealing. As a mild stimulant, it increases dopamine, thus giving patients the boost of dopamine they are used to from stimulants.
However, in studies of CUD treatment, modafinil has had mixed results. A 2017 meta-analysis of 11 studies with 896 participants found that modafinil wasn’t superior to placebo in improving treatment retention or achieving abstinence in CUD. However, a smaller subgroup analysis of six studies, conducted in the US, found that modafinil was superior to placebo in promoting cocaine abstinence (p = 0.035). The doses used in studies were well tolerated and varied between 200 mg daily and 400 mg daily, with most studies using 400 mg (Sangroula D et al, Subst Use Misuse 2017;52(10):1292–1306). High-dose modafinil is a reasonable option for stimulant use disorder, especially in patients with comorbid ADHD.
Stimulant substitution
Similar to methadone and buprenorphine for the treatment of opioid use disorder, and nicotine replacement for tobacco use, various psychostimulants have been tested as substitution therapy in patients with stimulant use disorder. These medications have similar mechanisms of action to the commonly misused stimulants but are safer and less habit-forming.
A 2016 Cochrane review examined psychostimulants and a few medications with stimulant-like effects in the context of CUD. It included 26 studies with 2,366 participants and assessed several medications: bupropion, dextroamphetamine, lisdexamfetamine, methylphenidate, modafinil, methamphetamine, mixed amphetamine salts, and selegiline. Results indicated that, as a group, psychostimulants decreased relapse rates in participants already abstinent from cocaine (p = 0.02), though there were problems with the quality of the studies. Psychostimulants didn’t reduce overall cocaine use or improve retention in treatment.
Looking at specific drugs, some stood out. Bupropion, dextroamphetamine, and mixed amphetamine salts were more efficacious than placebo in achieving sustained cocaine abstinence. Modafinil appeared to be more efficacious than placebo in reducing cocaine use. Lisdexamfetamine significantly decreased cocaine cravings compared to placebo.
However, since the number of studies investigating each drug was small, and treatment retention was a major issue, the overall quality of the results was compromised. While an intriguing option to keep an eye on, stimulant substitution isn’t quite ready for prime time, at least not as a first-line treatment (Castells X et al, Cochrane Database Sys Rev 2016(9):CD007380).
Approach
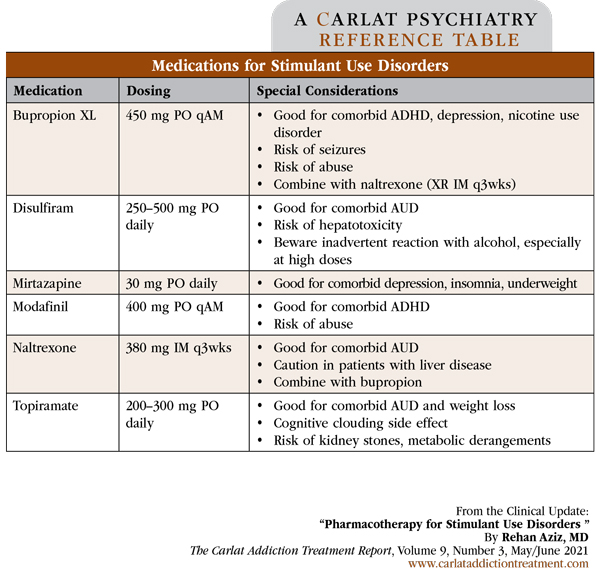
There are no clear-cut winners here, but based on the limited data we have and the safety profiles of the medications, the following could be considered for the treatment of stimulant use disorders:
Lisdexamfetamine is also an intriguing choice, though trials have used very high doses that, at this point, cannot be recommended for routine use.
CATR Verdict: The evidence base for the pharmacologic treatment of stimulant use disorders is mixed, with only a few positive trials for a variety of stimulants. Though there are a few promising contenders, medications still serve primarily in a backup role. Consider comorbidities before prescribing.
Addiction TreatmentMedications
Antidepressants
Antidepressants are thought to work in stimulant use disorder by treating stimulant-induced depression and withdrawal dysphoria. A Cochrane systematic review looked at the evidence in cocaine use disorder (CUD) by appraising 37 randomized controlled trials with 3,551 total participants. Studies included an array of antidepressants, most commonly bupropion, desipramine, and fluoxetine. Depression improved, but there was no reduction in cocaine use (Pani PP et al, Cochrane Database Syst Rev 2011(12):CD002950).
Although antidepressants are not particularly effective as a class in the management of stimulant use disorder, there is some evidence for mirtazapine in methamphetamine use disorder. Two placebo-controlled trials, both conducted in a population of men who have sex with men, demonstrated that 30 mg nightly dosing of mirtazapine modestly decreased methamphetamine use. The first study, which was relatively small at 56 participants, showed a decrease in methamphetamine-positive urine tox screens (p = 0.04) over 12 weeks when coupled with regular counseling (Colfax GN et al, Arch Gen Psych 2011;68(11):1168–1175). A larger, more recent study (n = 120) also showed a significant decrease in methamphetamine use that persisted out to 36 weeks (p = 0.02). Adherence in both studies was poor, however, and could present a challenge in the real world.
There might be new hope for bupropion in the treatment of methamphetamine use disorder as well. Recently, a large 12-week double-blind, placebo-controlled trial tested the combination of bupropion and naltrexone (Trivedi MH et al, N Engl J Med 2021;384(2):140–153). 628 adults received either extended-release injectable naltrexone (380 mg every 3 weeks) + oral extended-release bupropion (450 mg daily) or placebo. Patients who received the active drugs had fewer urine tox screens positive for methamphetamine than patients who received placebo, though the total number of negative tox screens was quite low in both groups (13.6% vs 2.5%). Mirtazapine and bupropion combined with naltrexone could be worth keeping in your back pocket. Give mirtazapine particular consideration if your patient struggles with insomnia or weight loss, which are both common in those who misuse stimulants.
Cocaine vaccine
Believe it or not, researchers have been developing a cocaine vaccine for years. The vaccine is supposed to work by producing cocaine-specific antibodies that block cocaine’s passage into the brain, thereby preventing the reinforcing effects of drug use. Although an intriguing idea, results were largely negative in two large trials (Martell BA et al, Arch Gen Psychiatry 2009;66(10):1116–1123; Kosten TR et al, Drug Alcohol Depend 2014;140:42–47). Research continues, but as of now, the cocaine vaccine is not ready for clinical use.
Disulfiram
Disulfiram isn’t just for alcohol use disorder (AUD) anymore—it holds promise for CUD, too. Recall that disulfiram causes an unpleasant reaction in patients who consume alcohol through its inhibition of acetaldehyde dehydrogenase. It also increases dopamine levels by blocking dopamine beta-hydroxylase. This could work to counter the dopamine depletion seen in chronic cocaine use and reduce cravings. Disulfiram also inhibits enzymes that metabolize cocaine. This leads to increased cocaine plasma levels, making cocaine use aversive.
Early studies with disulfiram were promising. However, it performed poorly in more recent studies, separating from placebo for length of cocaine abstinence and reduction in cocaine use in only one out of four trials. The studies, taken together, establish low-quality evidence to support the use of disulfiram (Ronsley C et al, PLoS ONE 2020;15(6):e0234809). You might keep it in mind, though, for patients who have failed other medications or for those with comorbid AUD.
GABAergic medications
GABA is the major inhibitory neurotransmitter of the CNS. By blocking activity of the dopamine reward system, GABAergic agents could prevent relapses by curbing stimulant-induced euphoria and reducing cravings. At least, that’s the theory.
Topiramate is the best studied and has yielded mixed results; out of five studies; only two were positive. Those two found that at doses of 200 mg and 300 mg daily, topiramate promoted abstinence in cocaine, amphetamine, and methamphetamine use disorders (Ronsley et al, 2020). Like disulfiram, topiramate may be especially helpful in patients with comorbid AUD.
Modafinil
The theoretical underpinning of using modafinil for stimulant use disorder is appealing. As a mild stimulant, it increases dopamine, thus giving patients the boost of dopamine they are used to from stimulants.
However, in studies of CUD treatment, modafinil has had mixed results. A 2017 meta-analysis of 11 studies with 896 participants found that modafinil wasn’t superior to placebo in improving treatment retention or achieving abstinence in CUD. However, a smaller subgroup analysis of six studies, conducted in the US, found that modafinil was superior to placebo in promoting cocaine abstinence (p = 0.035). The doses used in studies were well tolerated and varied between 200 mg daily and 400 mg daily, with most studies using 400 mg (Sangroula D et al, Subst Use Misuse 2017;52(10):1292–1306). High-dose modafinil is a reasonable option for stimulant use disorder, especially in patients with comorbid ADHD.
Stimulant substitution
Similar to methadone and buprenorphine for the treatment of opioid use disorder, and nicotine replacement for tobacco use, various psychostimulants have been tested as substitution therapy in patients with stimulant use disorder. These medications have similar mechanisms of action to the commonly misused stimulants but are safer and less habit-forming.
A 2016 Cochrane review examined psychostimulants and a few medications with stimulant-like effects in the context of CUD. It included 26 studies with 2,366 participants and assessed several medications: bupropion, dextroamphetamine, lisdexamfetamine, methylphenidate, modafinil, methamphetamine, mixed amphetamine salts, and selegiline. Results indicated that, as a group, psychostimulants decreased relapse rates in participants already abstinent from cocaine (p = 0.02), though there were problems with the quality of the studies. Psychostimulants didn’t reduce overall cocaine use or improve retention in treatment.
Looking at specific drugs, some stood out. Bupropion, dextroamphetamine, and mixed amphetamine salts were more efficacious than placebo in achieving sustained cocaine abstinence. Modafinil appeared to be more efficacious than placebo in reducing cocaine use. Lisdexamfetamine significantly decreased cocaine cravings compared to placebo.
However, since the number of studies investigating each drug was small, and treatment retention was a major issue, the overall quality of the results was compromised. While an intriguing option to keep an eye on, stimulant substitution isn’t quite ready for prime time, at least not as a first-line treatment (Castells X et al, Cochrane Database Sys Rev 2016(9):CD007380).
Approach
There are no clear-cut winners here, but based on the limited data we have and the safety profiles of the medications, the following could be considered for the treatment of stimulant use disorders:
- Modafinil
- Bupropion/naltrexone
- Mirtazapine
- Topiramate
- Disulfiram
Lisdexamfetamine is also an intriguing choice, though trials have used very high doses that, at this point, cannot be recommended for routine use.
CATR Verdict: The evidence base for the pharmacologic treatment of stimulant use disorders is mixed, with only a few positive trials for a variety of stimulants. Though there are a few promising contenders, medications still serve primarily in a backup role. Consider comorbidities before prescribing.
Table: Medications for Stimulant Use Disorder
(Click to view full-sized PDF.)
Issue Date: May 21, 2021
Table Of Contents
Recommended
Newsletters
Please see our Terms and Conditions, Privacy Policy, Subscription Agreement, Use of Cookies, and Hardware/Software Requirements to view our website.
© 2026 Carlat Publishing, LLC and Affiliates, All Rights Reserved.


_-The-Breakthrough-Antipsychotic-That-Could-Change-Everything.webp?t=1729528747)



