Home » PrEP: Introduction to the Basics
PrEP: Introduction to the Basics
November 22, 2019
From The Carlat Addiction Treatment Report
Great strides have been made in HIV treatment and prevention during the last 2 decades, resulting in falling numbers of new HIV infections during that time span. But since 2013, the rate of new infections has mostly plateaued at just under 40,000 new infections per year (www.hiv.gov). Those at highest risk of infection remain men who have sex with men, heterosexual persons with high-risk sexual behaviors, and persons who inject drugs. While many of us do not treat HIV infection, we can still be on the front lines of HIV prevention.
One primary prevention technique is PrEP, or pre-exposure prophylaxis—the daily use of the antiretroviral Truvada (tenofovir disoproxil 300 mg combined with emtricitabine 200 mg). The CDC now has guidelines calling for access to PrEP in primary care and addiction treatment settings (www.cdc.gov/hiv/pdf/risk/prep/cdc-hiv-prep-guidelines-2017.pdf). Although many clinicians are unfamiliar with Truvada, providing PrEP is straightforward and high-yield in addiction treatment settings. Here are the nuts and bolts of prescribing PrEP to your patients who are at high risk of HIV infection.
PrEP screening process
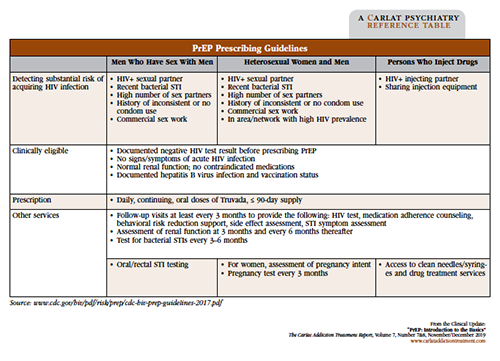
Begin by screening your patients who are most at risk. For people who inject drugs, you can ask, “In the past 6 months, have you injected by using needles, syringes, or other drug preparation equipment that had been used by another person?” Those with an injecting partner who is HIV+ are at highest risk. For all patients, make sure to get a complete sexual history, including frequency of condom use. Those at highest risk for HIV infection are homosexual or heterosexual persons who have an HIV+ partner, have a recent bacterial sexually transmitted infection (STI), have a high number of sexual partners, use condoms inconsistently or not at all, or participate in commercial sex work.
PrEP is only indicated for the prevention of HIV infection, and current HIV infection must first be ruled out, in part to prevent HIV drug resistance. While drug resistance from PrEP was not seen in clinical trials, experts will continue to monitor for emergent drug resistance as its use becomes more widespread. Serum or fingerstick HIV antibody/antigen testing is preferred—the rapid oral swab antibody testing is less sensitive (Stekler JD et al, J Clin Virol 2016;76:8–13). Acute HIV infection won’t show up on antibody testing, so ask about symptoms of infection in the past 4 weeks—most commonly fever, fatigue, myalgia, rash, and headache. Make sure renal function is normal and there is no bacterial STI present (chlamydia, gonorrhea, or syphilis). Other initial laboratory testing includes hepatitis B (HBV) serologies—all patients at high risk for HIV infection should be vaccinated for HBV. Truvada may also be used for HBV treatment, but this needs to go through a specialty provider.
How do you prescribe Truvada?
Once the screening process is complete, Truvada prescribing is straightforward. Getting therapeutic drug levels in the bloodstream will take up to 20 days, and for best protection, encourage consistent daily use—efficacy drops off once adherence falls below 85%. Only prescribe Truvada up to 90 days at a time, as HIV infection status needs to be rechecked every 3 months. At 6-month visits, order renal function and STI testing as well. Reevaluate high-risk behavior patterns regularly, and always provide information on safer sex and injection practices. Also, be sure to start patients on medications for opioid use disorder (OUD) or make appropriate referrals for that purpose.
Does PrEP work?
Several randomized, placebo-controlled trials informed the CDC guidelines and FDA recommendations, each looking at a particular population. In most trials, HIV infection rate was reduced by 50%–85%, varying with adherence rate. For people who inject drugs, the risk reduction was closer to the 50% mark with tenofovir disoproxil fumarate (TDF) 300 mg daily alone, not combined with emtricitabine, in a randomized controlled trial with open-label extension (Choopanya K et al, Lancet 2013;381(9883):2083–2090). Based on these data, the PrEP prescription for people who inject drugs can be either the Truvada combination pill or tenofovir alone. As of October 2019, Descovy, a combination of emtricitabine 200 mg and tenofovir alafenamide (TAF) 25 mg, was approved for PrEP as well, but only for use in men and transgender women. Truvada remains the only medication approved for PrEP use in all populations.
Side effects of Truvada are usually limited to the first month and are most commonly nausea, flatulence, and headache. There are no drug-drug interactions with the medications for OUD (buprenorphine, methadone, or extended release naltrexone). Do take note of any medications that may compromise kidney function, such as NSAIDs or lithium—monitor closely or avoid them altogether. If your patient is already on an antiretroviral therapy for another infection, refer to a specialist for PrEP evaluation.
Is PrEP feasible in non-ID settings?
For years, PrEP access was restricted to infectious disease (ID) specialists, presenting a major barrier. However, those on the front line—physicians in primary care and addiction treatment settings—encounter many more individuals who could benefit from HIV prophylaxis. In one study, nearly half of persons who injected drugs entering detoxification treatment were also willing to start on PrEP (Stein M et al, AIDS Behav 2014; 18(9):1694–1700). Yet, provider and staff comfort, as well as access to HIV lab testing, may influence the decision to offer PrEP. Providing training to addiction treatment providers as well as nursing and ancillary staff may help lower these perceived barriers. Pharmacies are also becoming potential access points for PrEP with the passage of a recent bill in California (www.tinyurl.com/yx2v9yaw). Lastly, it’s important to form relationships with ID providers so that you can easily refer anyone in need of HIV treatment.
CATR Verdict: PrEP with Truvada has proven efficacy in reducing new HIV infection rates. Clinicians working in addiction settings can offer protection with PrEP, combined with other risk reduction strategies. With a little knowledge and the right laboratory testing procedures, addiction treatment providers can be equipped to provide this high-yield intervention.
Addiction TreatmentOne primary prevention technique is PrEP, or pre-exposure prophylaxis—the daily use of the antiretroviral Truvada (tenofovir disoproxil 300 mg combined with emtricitabine 200 mg). The CDC now has guidelines calling for access to PrEP in primary care and addiction treatment settings (www.cdc.gov/hiv/pdf/risk/prep/cdc-hiv-prep-guidelines-2017.pdf). Although many clinicians are unfamiliar with Truvada, providing PrEP is straightforward and high-yield in addiction treatment settings. Here are the nuts and bolts of prescribing PrEP to your patients who are at high risk of HIV infection.
PrEP screening process
Begin by screening your patients who are most at risk. For people who inject drugs, you can ask, “In the past 6 months, have you injected by using needles, syringes, or other drug preparation equipment that had been used by another person?” Those with an injecting partner who is HIV+ are at highest risk. For all patients, make sure to get a complete sexual history, including frequency of condom use. Those at highest risk for HIV infection are homosexual or heterosexual persons who have an HIV+ partner, have a recent bacterial sexually transmitted infection (STI), have a high number of sexual partners, use condoms inconsistently or not at all, or participate in commercial sex work.
PrEP is only indicated for the prevention of HIV infection, and current HIV infection must first be ruled out, in part to prevent HIV drug resistance. While drug resistance from PrEP was not seen in clinical trials, experts will continue to monitor for emergent drug resistance as its use becomes more widespread. Serum or fingerstick HIV antibody/antigen testing is preferred—the rapid oral swab antibody testing is less sensitive (Stekler JD et al, J Clin Virol 2016;76:8–13). Acute HIV infection won’t show up on antibody testing, so ask about symptoms of infection in the past 4 weeks—most commonly fever, fatigue, myalgia, rash, and headache. Make sure renal function is normal and there is no bacterial STI present (chlamydia, gonorrhea, or syphilis). Other initial laboratory testing includes hepatitis B (HBV) serologies—all patients at high risk for HIV infection should be vaccinated for HBV. Truvada may also be used for HBV treatment, but this needs to go through a specialty provider.
How do you prescribe Truvada?
Once the screening process is complete, Truvada prescribing is straightforward. Getting therapeutic drug levels in the bloodstream will take up to 20 days, and for best protection, encourage consistent daily use—efficacy drops off once adherence falls below 85%. Only prescribe Truvada up to 90 days at a time, as HIV infection status needs to be rechecked every 3 months. At 6-month visits, order renal function and STI testing as well. Reevaluate high-risk behavior patterns regularly, and always provide information on safer sex and injection practices. Also, be sure to start patients on medications for opioid use disorder (OUD) or make appropriate referrals for that purpose.
Table: PrEP Prescribing Guidelines
Click to view as full-size PDF.
Does PrEP work?
Several randomized, placebo-controlled trials informed the CDC guidelines and FDA recommendations, each looking at a particular population. In most trials, HIV infection rate was reduced by 50%–85%, varying with adherence rate. For people who inject drugs, the risk reduction was closer to the 50% mark with tenofovir disoproxil fumarate (TDF) 300 mg daily alone, not combined with emtricitabine, in a randomized controlled trial with open-label extension (Choopanya K et al, Lancet 2013;381(9883):2083–2090). Based on these data, the PrEP prescription for people who inject drugs can be either the Truvada combination pill or tenofovir alone. As of October 2019, Descovy, a combination of emtricitabine 200 mg and tenofovir alafenamide (TAF) 25 mg, was approved for PrEP as well, but only for use in men and transgender women. Truvada remains the only medication approved for PrEP use in all populations.
Side effects of Truvada are usually limited to the first month and are most commonly nausea, flatulence, and headache. There are no drug-drug interactions with the medications for OUD (buprenorphine, methadone, or extended release naltrexone). Do take note of any medications that may compromise kidney function, such as NSAIDs or lithium—monitor closely or avoid them altogether. If your patient is already on an antiretroviral therapy for another infection, refer to a specialist for PrEP evaluation.
Is PrEP feasible in non-ID settings?
For years, PrEP access was restricted to infectious disease (ID) specialists, presenting a major barrier. However, those on the front line—physicians in primary care and addiction treatment settings—encounter many more individuals who could benefit from HIV prophylaxis. In one study, nearly half of persons who injected drugs entering detoxification treatment were also willing to start on PrEP (Stein M et al, AIDS Behav 2014; 18(9):1694–1700). Yet, provider and staff comfort, as well as access to HIV lab testing, may influence the decision to offer PrEP. Providing training to addiction treatment providers as well as nursing and ancillary staff may help lower these perceived barriers. Pharmacies are also becoming potential access points for PrEP with the passage of a recent bill in California (www.tinyurl.com/yx2v9yaw). Lastly, it’s important to form relationships with ID providers so that you can easily refer anyone in need of HIV treatment.
CATR Verdict: PrEP with Truvada has proven efficacy in reducing new HIV infection rates. Clinicians working in addiction settings can offer protection with PrEP, combined with other risk reduction strategies. With a little knowledge and the right laboratory testing procedures, addiction treatment providers can be equipped to provide this high-yield intervention.
KEYWORDS addiction addiction-treatment hiv medical_comorbidities medication pharmacology prevention substance-use substance-use-disorders
Issue Date: November 22, 2019
Table Of Contents
Recommended
Newsletters
Please see our Terms and Conditions, Privacy Policy, Subscription Agreement, Use of Cookies, and Hardware/Software Requirements to view our website.
© 2026 Carlat Publishing, LLC and Affiliates, All Rights Reserved.


_-The-Breakthrough-Antipsychotic-That-Could-Change-Everything.webp?t=1729528747)



